Gregarine Parasitism of Clams and Cockles
On this page
Category
Category 4 (Negligible Regulatory Significance in Canada)
Common, generally accepted names of the organism or disease agent
Parasitism by gregarines, Gymnospore infections of clams, Apicomplexan parasitism.
Scientific name or taxonomic affiliation
- Nematopsis veneris, Nematopsis ostrearum, Nematopsis schneideri, Nematopsis sp. and probably other species of the family Porosporidae (class Conoidasida, suborder Septatorina).
- Heteroxenous apicomplexan parasite with zoites that have some ultrastructural similarities to merozoites of Aggregata eberthi (class Coccidia, suborder Eimerioria).
Geographic distribution
- Probably ubiquitous, although each species may have a confined distribution
- Coast of British Columbia, Canada
Host species
- Cerastoderma (=Cardium) edule, Ruditapes decussatus, Cardium lamarcki, Saxicava rugosa, Tellina spp., Venerupis (=Tapes) philippinarum, Protothaca staminea, Nuttalia obscurata, Clinocardium nuttali and Saxidomus gigantea as well as other marine bivalves including oysters, mussels and scallops.
- Protothaca staminea. Rarely observed in Venerupis (=Tapes) philippinarum from the northern end of their distribution in British Columbia.
Impact on the host
- The gymnospores and oocysts (gregarine spores containing one or more uninucleate vermiform sporozoites), or naked sporozoites (depending on the species involved) usually occur within a phagocyte that can move within the connective tissue to most organs but are most frequently observed in the gills. Infection is usually associated with a focal, benign accumulation of a several haemocytes, without significant health effects. Although prevalence can be high, the intensity of infection is usually too low to compromise the health of the clam. For example, a mean prevalence of 76% occurred in C. edule populations from all 34 locations examined along the coast of Galicia, Spain but the intensity of infection was usually low (few Nematopsis oocysts per cockle) (Carballal et al. 2001). Multiplication of gregarines is limited in bivalves with the completion of the life cycle occurring in the lumen of the intestinal tract of marine arthropods.
- High prevalence (usually between 70 and 100 %) of P. staminea with cysts in the connective tissue. Although the cyst (containing numerous closely packed zoites) can be up to 150 µm in diameter, there was little evidence of a haemocytic response to infection and no associated pathology. The cysts probably represent a stage of a heteroxenous coccidian life cycle, with a predator of clams serving as the definitive host in which gamagony and sporogony occur.
Diagnostic techniques
Squash Preparations
- Because of the larger sample size that can be examined in comparison to histology, microscopic examination (100 x) of gills squashed between glass slides is the most sensitive procedure for detecting the oval oocysts with their light refractile walls. However, the gymnospores and naked sporozoites may not be detected by this technique.
- Small pieces of clam body tissue pressed between a glass slide and coverslip and examined under a compound microscope. Cysts can be ruptured by applying pressure to the coverslip to free the zoites which exhibit flexing and gliding movements. Each elongate zoite (about 25 by 4 µm) has a nipple-like protrusion at the anterior end.
Histology
- All stages that occur in bivalves can be observed in the connective tissue of various organs but they are most prevalent in the gills. However, the presence of these parasites can be missed easily due to the normally low intensity of infection.
- Cysts, ranging in size from 20 to 150 µm and irregular in shape, occur in the connective tissue, usually around the intestine and kidney. The cysts are thick walled (about 4 µm) and all contain numerous mature banana-shaped zoites. Although cysts of various sizes were observed in clams collected at different times of the year, earlier stages of development could not be identified with certainty. There is little evidence of a haemocyte aggregation response to the cysts in any of the tissues.

Figure 1. Several longitudinal and cross sections through Nematopsis sp. spores (arrows) in the connective tissue of the gills of a butter clam, Saxidomus gigantea. Despite the heavier than normal infection in this clam, there is no cellular response to the infection by the clam. Hematoxylin and eosin stain.

Figure 2. Higher magnification of a Nematopsis sp. spore (arrow) in the connective tissue of the gill of a Pacific littleneck clam, Protothaca staminea. Hematoxylin and eosin stain.

Figure 3. Cysts (arrows) of the apicomplexan parasite in the connective tissue around the gut of a Pacific littleneck clam, Protothaca staminea. Hematoxylin and eosin stain.
Electron Microscopy
- The monozoic, ellipsoial oocyst (13.3 µm by 4.5 µm) has a dense wall (about 0.35 µm thick) and contains a single vermiform uninucleate sporozoite. The apical zone of the wall contains a circular micropyle covered by a conical operculum. Externally the wall was surrounded by numerous thick and long microfibrils attached to the wall and connected to the host cell (Azevedo and Cachola 1992).
- The cyst wall appeared homogeneously electron dense. The external surface of the cyst wall was highly irregular and exhibited numerous small projections that were surrounded by small dense particles in the zone between the cyst and host tissue. The latter was separated from the parasite by a pronounced striated zone adjacent to labyrinthine structures. The zoites lay within the cyst in a electron-lucent fibrillar/granular matrix. The apical region of the zoite was heavily reinforced with an electron-dense, cap-like structure. A dense cylindrical collar, thickened apically and tapering posteriorly, lay beneath the apex of the zoite and appeared to be the origin of about 55 posteriorly radiating pellicular microtubules. Polar rings were absent and appeared to be replaced by the apical dense cylindrical collar. Within this apical collar lay a typical conoid through which ducts of the elongated rhoptries and small micronemes appeared to extend. The cytoplasm of the zoites contained numerous amylopectin inclusions similar to those of Aggregata eberthi.
Figures 4 to 7. Electron micrographs illustrating ultrastructural features of the cystic stages of the apicomplexan parasite in the connective tissue of the digestive gland of a Pacific littleneck clam, Protothaca staminea. Tissue was post fixed in osmium tetroxide and stained with lead citrate. Images courtesy of S.S. Desser and H. Hong as published by Desser and Bower (1997).

Figure 4. Portion of a cyst showing zoites (Zo, Nu = nucleus), the thick, homogeneously dense cyst wall (Cw), and adjacent host tissue (asterisk). The host-cell boundary layer consists of a striated zone (Sz), underlain by a labyrinthine structure (Mv).
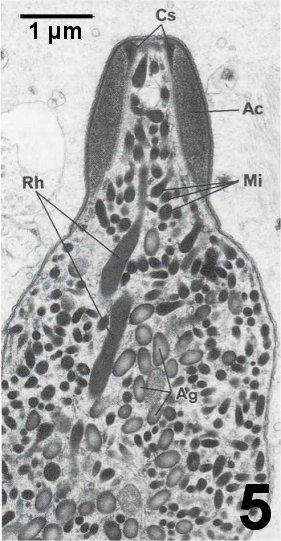
Figure 5. Longitudinal section through anterior end of zoite showing thickened dense apical cap region (Ac). Note the apical dense cylindrical structure (Cs), which tapers posteriorly. Elongated rhoptries (Rh) and smaller dense micronemes (Mi) are apparent. The cytoplasm contains numerous amylopectin granules (Ag). The matrix in the cyst consists of lucent granular and fibrillar material and vesicular inclusions.

Figure 6. Longitudinal section through apical dense cylindrical structures (Cs) with posteriorly radiating pellicular microtubles.
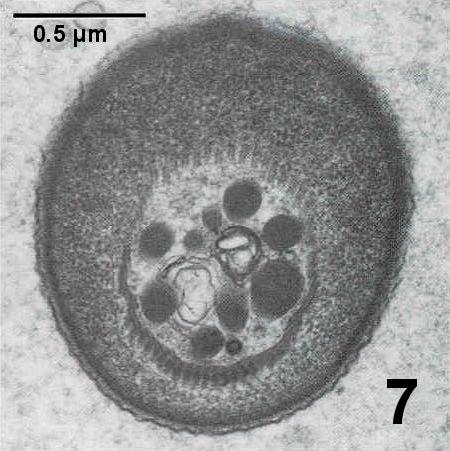
Figure 7. Oblique section through dense apical cap region illustrating the approximately 55 pellicular microtubles.
Methods of control
For Nematopsis sp., the source of the shrimp seed may be an important factor because wild post larvae had higher survival and lower prevalence of infection then laboratory reared post larvae. Medicated feeds that were tested did not alleviate the infection or improve survival. The source of P. scolecoides was not identified but the shrimp acquired the infection at the culture facility. Because gregarines require at least two hosts (usually a mollusc or annelid worm in addition to the crustacean) to complete their life cycle, infection can be circumvented by removal of the alternate host(s) from the culture facility or water source.
References
Azevedo, C. and R. Cachola. 1992. Fine structure of the apicomplexa oocyst of Nematopsis sp. of two marine bivalve molluscs. Diseases of Aquatic Organisms 14: 69-73.
Bower, S.M., J. Blackbourn and G.R. Meyer. 1992. Parasite and symbiont fauna of Japanese littlenecks, Tapes philippinarum (Adams and Reeve, 1850), in British Columbia. Journal of Shellfish Research 11: 13-19.
Carballal, M.J., D. Iglesias, J. Santamarina, B. Ferro-Soto and A. Villalba. 2001. Parasites and pathologic conditions of the cockle Cerastoderma edule populations of the coast of Galicia (NW Spain). Journal of Invertebrate Pathology 78: 87-97.
Desser, S.S. and S.M. Bower. 1997. The distribution, prevalence, and morphological features of the cystic stage of an apicomplexan parasite of native littleneck clams (Protothaca staminea) in British Columbia. The Journal of Parasitology 83: 642-646.
Lauckner, G. 1983. Diseases of Mollusca: Bivalvia. In: O. Kinne (ed.). Diseases of Marine Animals. Volume II: Introduction, Bivalvia to Scaphopoda. Biologische Anstalt Helgoland, Hamburg, p. 542-548.
MacCallum, G.S., J. Blackbourn, S.E. McGladdery, S.M. Bower and J.T. Davidson. 2001. Disease issues relevant to the culture of shellfish in Atlantic and Pacific Canada. Bulletin of the Aquaculture Association of Canada 101-3: 5-12.
Marshall, W.L., S.M. Bower and G.R. Meyer. 2003. A comparison of the parasite and symbiont fauna of cohabiting native (Protothaca staminea) and introduced (Vennerupis philippinarum and Nuttalia obscurata) clams in British Columbia. Journal of Shellfish Research 22: 185-192.
Citation Information
Bower, S.M., (2007): Synopsis of Infectious Diseases and Parasites of Commercially Exploited Shellfish: Gregarine Parasitism of Clams and Cockles.
Date last revised: October 2007
- Date modified: