Carrying capacity modeling for bivalve aquaculture: Invasive tunicates and biodeposition
Issue 3 : April 2012
Summary
Production from suspended bivalve aquaculture has increased significantly over the past two decades, but recently has been impacted negatively by significant fouling from invasive tunicate species. Although invasive tunicates can represent a substantial biomass on suspended culture gear, there are no direct data available regarding biodeposition from these fouling communities. Research has suggested that sedimentation from suspended bivalves in culture and the associated fauna may contribute considerably to the total amount of material that settles to the bottom. The following research was done under field conditions to establish the extent to which fouling organisms contribute to biodeposition from mussel culture.
The biodeposition dynamics associated with mussels and two fouling tunicates, Ciona intestinalis and Styela clava, in mussel aquaculture in Prince Edward Island (PEI) were examined in this study. The presence of C. intestinalis on mussel socks doubled the amount of biodeposition relative to mussel socks without tunicates. Although S. clava, used in the experiment, were small and had a negligible effect on total biodeposition from mussel socks, they increased sedimentation rates from mussel socks relative to that of abiotic control socks. Sinking rates of faecal pellets from large C. intestinalis varied between 1.4 and 6.5 cm s-1. Using rates of biodeposit production and sinking, and hydrological data obtained from the present study, footprints of benthic loading due to mussel and tunicate biodeposition for a typical mussel farm in PEI were modeled using Shellfish-DEPOMOD. This model predicted benthic loading below mussel socks fouled with C. intestinalis to be approximately two times greater than that from lines containing only mussels − rates of up to 15.2 g m-2 d-1− which is in agreement with the research study results. However, the extent of the biodeposition footprint (≥ 1 g m-2 d-1) below C. intestinalis-fouled mussel socks is similar to or more confined compared to mussel only socks due the greater settling rate of C. intestinalis biodeposits.
Results from this research will increase our understanding of aquaculture-environmental interactions and help improve overall environmental management of benthic impacts of shellfish aquaculture in Canadian waters.
Introduction

A. Unfouled mussel sock
B. Mussel sock fouled by C. intestinalis and S. clava
C. Styela clava
D. Ciona intestinalis
Suspended bivalve aquaculture production has increased significantly over the past twenty years. This form of production has well-documented influences on underlying infaunal benthic communities (Mattsson and Lindén 1983, Callier et al. 2007). The general response of affected benthic communities to such organic loading includes decreased species richness and a possible increase in the individual abundance of a few opportunistic species (including increased biomass), reduced overall community biomass, a general or species-specific decrease in body size, a shallowing of the portion of the sediment column occupied by infauna, and a shift in the relative dominance of trophic groups (Pearson and Rosenberg 1978, Callier et al. 2008). Ultimately, this may also include sediment anoxia and, in the most severe of cases, a dominance by microbial mats.
Recently, a number of invasive tunicates have become important fouling organisms in suspended bivalve culture areas throughout the world (Lambert 2007, McKindsey et al. 2007). When these animals become associated with suspended bivalve culture, they become a serious concern for the industry (Enright 1993). Their presence on cultured suspended bivalves can have an important influence on fluxes of organic matter to the bottom and consequent impacts on benthic communities. As the magnitude of benthic loading determines the magnitude of benthic infaunal responses (Callier et al. 2009), knowledge of such rates is important for predicting suspended bivalve culture impacts on the benthic environment (Weise et al. 2009). The scale of the increase in benthic loading associated with fouling by tunicates in aquaculture remains largely unstudied.
Our research on bivalve carrying capacity modeling has found that realistic benthic community changes can be predicted in some cases with the use of the particle tracking model, Shellfish-DEPOMOD (Weise et al. 2009). In those cases where such predictions are unrealistic, it is, in part, because biodeposition from the fouling organisms associated with mussels in culture has not been considered. Fouling organisms can represent a considerable biomass that, in the case of tunicates, may be several times that of the mussels to which they are attached. To improve the applicability of results from modeling, we require realistic estimates of biodeposit production (i.e., faeces) and sedimentation (i.e., sinking speed).
The objective of this PARR research project was to establish the extent to which fouling organisms affect biodeposition for mussel culture under field conditions. Specifically, this study examined the influence of tunicates growing on suspended mussel aquaculture “socks” (individual polyethylene sleeves of mussels suspended from subsurface longlines) on the loading of biodeposits to the bottom. We compared how the presence of the two fouling solitary tunicates, Ciona intestinalis and Styela clava, influence the production of biodeposits from mussel socks. This study evaluated biodeposit production by small mussel socks that were constructed for this purpose under field conditions. It also measured the sinking velocity of tunicate (C. intestinalis) faeces and compared predicted benthic loading footprints around farms with and without fouling tunicates using the results from the present study within a hydrodynamic-based particle dispersion model. As this research was conducted under field conditions, results are readily transferable to Shellfish-DEPOMOD modeling exercises.
Methods
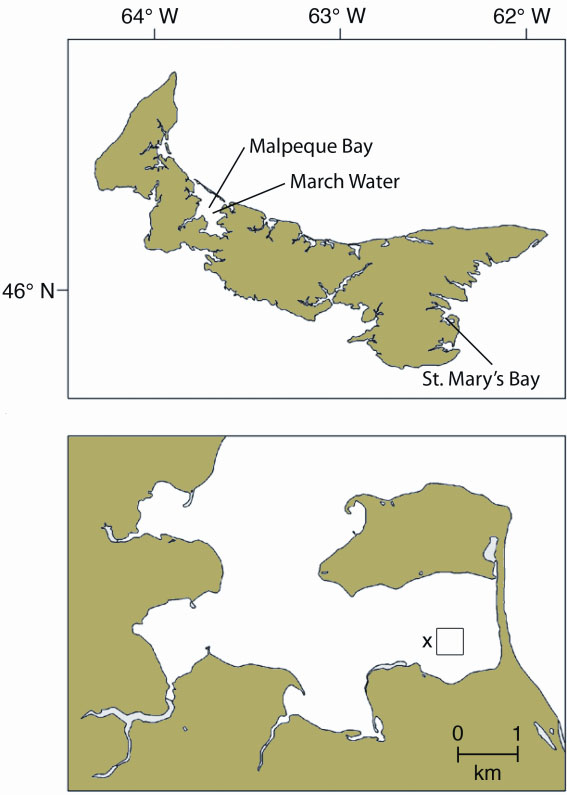
Figure 1. Location of the study sites in Prince Edward Island (top), and, in closer detail, St. Mary's Bay (below). The “x” represents the location of the current profiler used to measure hydrodynamic characteristics of the sampling area. The square represents the lease-site where the biodeposit production experiments were done.
This study was carried out in the March Water area of Malpeque Bay and in St. Mary's Bay, PEI (Figure 1). A total of 36 mussel socks (40 cm long) and an equivalent number of control socks (inert shell equivalents) were deployed. Half of each of these were placed on a commercial mussel line at ca. 3 m depth in each of March Water (site dominated by Styela) and St. Mary's Bay (site dominated by Ciona) to allow tunicates to settle and grow on the socks in late June, 2008. Socks from March Water were transferred to St. Mary's Bay in mid-October to evaluate biodeposition.
Biodeposit production was measured by placing individual mussel socks over/within large sediment traps (Figure 2) and collecting sedimented material after one day. The percent organic matter of the sedimented material samples was determined by calculating the weight loss of samples dried at 65 °C and the same ones combusted at 450 °C for 5 h. Methods similar to those of Callier et al. (2006) were used to evaluate Ciona biodeposit sinking velocities. Sinking velocity trials for Styela faecal pellets were not performed as Styela faeces could not be accurately identified from the sedimented material.
Biodeposition from suspended mussel lines with and without fouling organisms was modeled for a hypothetical shellfish farm in PEI using Shellfish-DEPOMOD (Weise et al. 2009) and the biodeposition data from this study. Shellfish-DEPOMOD is a hydrodynamic-based particle dispersion model that consists of four modules that are applied consecutively: grid generation, particle tracking, resuspension, and benthic impact.
Results
The mean biomass of Ciona was greater on empty control socks than on mussel socks, although the abundance was similar. Of the original 100–150 mussels per sock, a mean of 45 mussels per sock were alive in October; as the empty shells were still present, most had presumably smothered under the sleeve of Ciona that had developed around the socks. The final abundance of mussels in socks fouled by Styela in Malpeque was about 3 times that of Ciona socks from St. Mary's Bay despite similar initial stocking numbers. The late recruitment and the small size of Styela (mostly less than 5 cm long) limited the use of data for this species to comparison purposes only. The focus of this study is primarily on the Ciona data.
Sedimentation associated with socks on which Ciona attached was approximately 14 g (dry weight) trap-1 d-1 greater than background (3.3 g trap-1 d-1) and did not differ between socks with mussels or the controls. Sedimentation associated with mussels with Styela attached or removed was approximately 6–7 g trap-1 d-1 greater than background rates. Ciona presence on socks was also shown to have resulted in significantly greater organic biodeposit enrichment compared to either the control or Styela-fouled socks. However, the proportion of organic matter below those mussel socks on which Styela had attached did not differ from that found below those mussel socks which had tunicates removed, or from that observed below the control socks.

Figure 2. Underwater photograph of experimental sediment traps used in St. Mary's Bay. The white box indicates a mussel sock fouled with the invasive tunicate (Ciona intestinalis). Inset image shows a typical mussel sock without fouling from invasive tunicates.
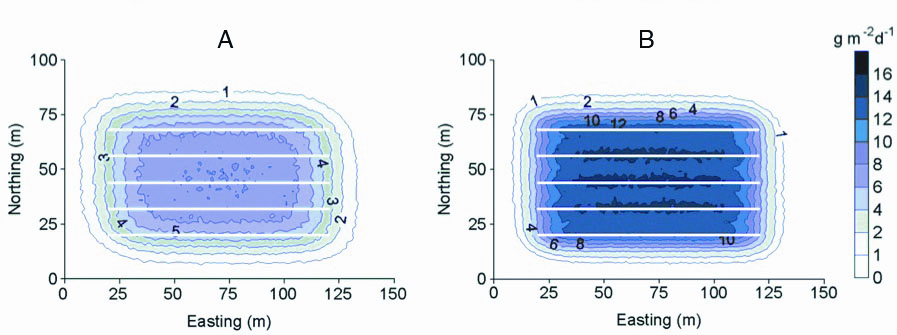
Figure 3. Modeled biodeposition footprint (g m-2d-1, dry weight) below an idealized Prince Edward Island suspended mussel farm with combinations of mussels and fouling tunicates: A) mussels only, B) mussels and Ciona intestinalis. Biodeposition rates (indicated with the legend on the right and contour line markers on each plot) were modeled for 5 backlines (white lines on each plot) measuring 100 m each and based on currents measured over a 24 h period in St. Mary's Bay, PEI. (after McKindsey et al. 2009).
These biodeposition values were then fed into the Shellfish-DEPOMOD model to simulate farm-scale biodeposition (g m-2 d-1). The model predicted biodeposition rates of up to 7.3 g m-2 d-1 directly beneath “mussels only” lines and a deposition gradient extending out with increasing distance from the lines (Figure 3-A). The predicted deposition sphere extended out approximately 15–20 m from the lines. In contrast, deposition directly below lines with mussels and Ciona demonstrated rates of up to 15.2 g m-2 d-1, approximately 2 times greater than that from lines with only mussels (Figure 3-B). However, given the greater settling rate of Ciona biodeposits relative to mussel biodeposits, the extent of the modeled benthic deposition footprint is similar to or more confined compared to mussel only socks.
Conclusion
Overall, biodeposit production was within the broad range reported previously in other studies (see review in Weise et al. 2009). Bivalves in suspended culture shunt organic material to the bottom at rates much greater than background levels and influence benthic communities in a manner consistent with the organic enrichment model of Pearson and Rosenberg (1978) (Figure 4).
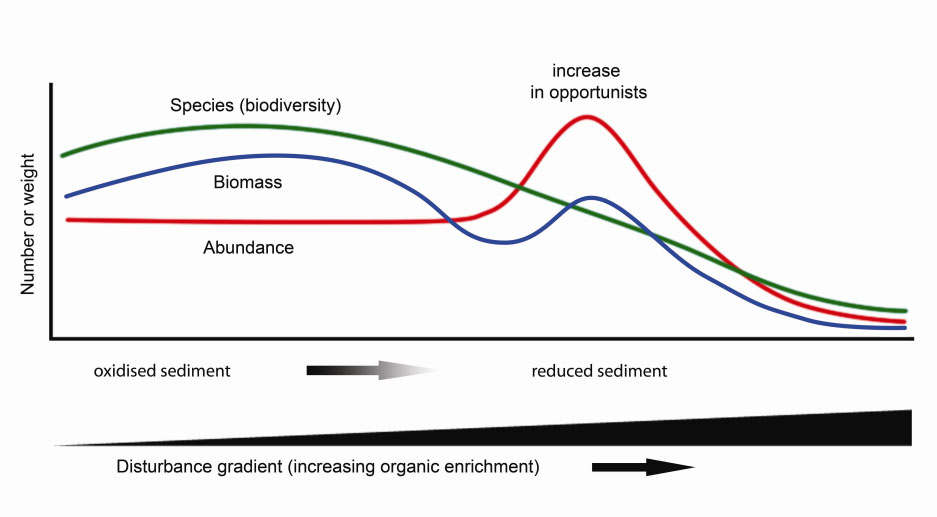
Figure 4. Generalized patterns of benthic invertebrate community and sediment changes as they are related to benthic disturbance and organic enrichment (after Nilsson and Rosenberg (2000)).
Although the contribution of associated organisms (including invasive tunicates) to the flux of organic matter to the bottom [biodeposition] has long been recognized as a consequence of suspended bivalve culture (Dahlbäck and Gunnarsson 1981, Mattsson and Lindén 1983), it has remained largely unstudied. Biodeposition rates below Ciona-fouled mussel socks was double that found below Styela-fouled socks even though mussel biomass on Styela-fouled socks was three times that of the Ciona-fouled socks.
The sinking rate of Ciona faeces was found to be about two to three times that reported for Mytilus edulis (Blue Mussel) of a commercial size or less, and greater than that reported for other species of mussels (Callier et al. 2006). Accordingly, although Ciona increases overall biodeposit production, the greater sinking velocity of Ciona faecal pellets will act to restrain the area of the biodeposition footprint to the bottom directly below the mussel lines, although at greatly increased concentration.
This research has shown that biodeposition from mussel socks fouled with Ciona was approximately two times that of unfouled socks and about five times that of natural background. It was also shown that organic loading-related effects are more severe and localized when mussel socks were fouled by Ciona. These conclusions were supported by the Shellfish-DEPOMOD modeling exercise which also showed that organic loading was expected to be concentrated in areas directly beneath the mussel lines. This was partially a reflection of the weak currents measured at the study site (Figure 1). The model assumed that resuspension of biodeposits was negligible but for areas that have stronger currents, the resulting benthic footprint may end up being larger.
References
- Callier, M.D., C.W. McKindsey, and G. Desrosiers. 2007. Marine Ecology Progress Series 348:103–115.
- Callier, M.D., C.W. McKindsey, and G. Desrosiers. 2008. Aquaculture 278:77–88.
- Callier, M.D., et al. 2006. Marine Ecology Progress Series 322:129–141.
- Callier, M.D., et al. 2009. Marine Pollution Bulletin 58:1544–1553.
- Dählback, B. and L. Å.H. Gunnarsson. 1981. Marine Biology 63:269–275.
- Enright, C. 1993. World Aquaculture 24:44–46.
- Lambert, G. 2007. Jounal of Experimental Marine Biology and Ecology 342:3–4.
- Mattsson, J. and O Lindén. 1983. Sarsia 68:97–102.
- McKindsey, C.W., et al. 2007. Journal of Shellfish Research 26:281–294.
- McKindsey, C.W., et al. 2009. Aquaculture 295:44-51.
- Nilsson, H.C. and R. Rosenberg. 2000. Marine Ecology Progress Series 197:139–149.
- Pearson, T.H. and R. Rosenberg. 1978. Oceanography and Marine Biology Annual Review 16:229–311.
- Weise, A.M., et al. 2009. Aquaculture 298:239–253.
The lead scientist on this PARR project (PARR-2008-QC-09), Dr. Chris McKindsey, can be contacted at Chris.McKindsey@dfo-mpo.gc.ca.
Published by: Fisheries and Oceans Canada Aquaculture Science Branch Ottawa, ON K1A 0E6
©Her Majesty the Queen in Right of Canada 2012
DFO/2012-1807
Cat. No. Fs45-2/2012E (Print)
ISBN 978-1-100-20171-9 (Print)
ISSN 1929-7939 (Print)
Cat. No. Fs45-2/2012E-PDF (Online)
ISBN 978-1-100-20172-6 (Online)
ISSN 1928-1013 (Online)
- Date modified: