Aquaculture Collaborative Research and Development Program (ACRDP) Fact Sheet
Issue 17 - March, 2013
Summary
Signal Crayfish (Pacifastacus leniusculus) are thought to be a species of commercial production potential, however there are a number of culture techniques that need to be better refined before commercial production is viable in Canada. This project sought to establish culture techniques for the species, by assessing growth and survival of juveniles as well as the digestive capabilities of adult crayfish fed formulated diets containing sustainable feed ingredients. The sustainable ingredients chosen were soybean meal, a common feed substitute for fishmeal, and Duckweed (Spirodela polyrrhiza), an invasive freshwater plant species, in British Columbia. During this project, Signal Crayfish were successfully raised for an entire year with minimal mortalities. Each of the diets tested showed very good survival and similar levels of growth. Crayfish consumed higher proportions of the Duckweed diet despite a slightly lower digestibility which suggests that aquatic macrophytes may be important feed ingredients for increasing palatability and caloric intake. This work provided a key first step in the culture of Signal Crayfish in British Columbia, and has shown that it is possible to breed and rear Signal Crayfish in captivity.
The Aquaculture Collaborative Research and Development Program (ACRDP) is a Fisheries and Oceans Canada (DFO) initiative to increase the level of collaborative research and development activity between the aquaculture industry and DFO.

Figure 1
Adult females brooding eggs, housed singly in a vertical incubator divided into separate mesh-bottom compartments that allowed recently released juveniles to fall through, preventing cannibalism (Photo: Anya Dunham, DFO)Introduction
Signal Crayfish (Pacifastacus leniusculus) are a highly-valued farmed species in both foreign and domestic markets. Although this species is cultured in foreign countries such as Sweden (where it is considered an invasive species), there have been few attempts to raise Signal Crayfish within their native range (i.e., Canada to northwestern United-States). Developing techniques for raising Signal Crayfish represents a critical first step in developing this industry in British Columbia. However, reliable data from laboratory experiments on growth and survival rates are essential for the development of sound business models for culture operations. Furthermore, there is a growing trend towards developing and utilizing ecologically-sustainable feed ingredients for aquaculture species. Given their omnivorous feeding habits, it may be possible to raise Signal Crayfish on diets primarily composed of sustainable ingredients. Therefore understanding the nutritional requirements of Signal Crayfish is not only important for optimizing growth, but also for increasing consumer acceptance of a new species in local markets.
The goals of this project were: 1) to establish successful culture techniques for the development of Signal Crayfish from egg to adult stage; 2) to determine growth and survival in the laboratory for juvenile crayfish raised on a reference (fishmeal) diet, and diets composed of large proportions of ingredients considered to be more sustainable (soybean meal and Duckweed or Spirodela polyrrhiza*); and, 3) to determine the digestibility, and therefore the potential effectiveness, of sustainable feed ingredients in prepared diets for adult Signal Crayfish.
* Duckweed is an invasive species to British Columbia but found in great abundance in the southwest of the province in water bodies with high nutrient loads (such as golf course ponds).
Methods
Berried female broodstock of P. leniusculus (average carapace length: 45.0 ± 1.3 mm) was acquired from Asturia Aquaculture Crayfish Consulting on March 31, 2011. These animals were wild broodstock that had bred while in captivity. Broodstock were held in vertical incubators, with each tray divided into 6 separate compartments by plastic dividers, each containing a single berried female (Fig. 1).
The bottom of each compartment consisted of 10 mm mesh, allowing juveniles to fall through once they became free living (i.e., following the transition from stage I to stage II). This helped to minimize cannibalism of the juveniles by the adult broodstock. The trays were provided with aerated, dechlorinated municipal water at 15°C at a flow rate of 5 L/min. To maintain pH and calcium hardness at levels that were conducive to growth, water was passed through a column containing crushed oyster shell before entering the tank. Using this method, pH values were maintained between 7.2 and 7.5, and calcium hardness was maintained at about 24 ppm. Lighting was provided solely by overhead fluorescent lights, and was maintained on a 12 h light: 12 h dark cycle. Broodstock were fed daily on a diet of pelleted salmon feed until the appearance of the first stage I juveniles, after which they were starved. This ensured that the juveniles were not exposed to exogenous sources of food prior to administration of test diets.
To determine the effect of various diets on growth and survival, stage II juvenile crayfish were randomly allocated to trays in a vertical incubator as described above, with the following exceptions: each tray was divided into 20 separate compartments by clear acrylic dividers; and, each compartment contained an individual juvenile crayfish.
The top and bottom of each chamber were lined with 1 mm mesh to allow water flow. This presumably allowed the crayfish to see each other, but prevented physical interaction and cannibalism. Each compartment was randomly assigned one of 3 dietary treatments (n = 6 per diet, per tray) or starved as controls (n = 2 per tray). Diet treatments were composed as follows: 1) the reference diet (REF) using fishmeal as the protein source; 2) the Duckweed diet (DW) composed of 30 % Duckweed, and 70 % reference diet; and, 3) the soybean meal diet (SBM) composed of 30 % soybean meal, and 70 % reference diet (Table 1). Each tray contained an equal number of each diet treatment to minimize any possible effects of tray placement within the vertical incubator. A total of 140 individuals were tested in seven trays. Every weekday morning for one year, excess food was removed from each chamber and animals (except for starved controls) had free access to their assigned test diet. During this time, any mortalities or moults were recorded and removed. Each month, the crayfish were weighed and measured.
Table one has four columns depicting the amount of ingredients by kilogram weight in three diet formulations: a reference diet, a Duckweed diet and a soybean meal diet. The first column lists nine measured ingredients in the diet. The second column shows the amount of each ingredient in the reference diet. The third column shows the amount of each ingredient in the Duckweed diet. The fourth column shows the amount of each ingredient in the soybean meal diet.
| Ingredient | REF | DW | SBM |
|---|---|---|---|
| Wheat Flour | 340.0 | 238.0 | 238.0 |
| Fish Oil | 20.0 | 14.0 | 14.0 |
| Fish Meal | 540.0 | 378.0 | 378.0 |
| Duckweed | 0.0 | 300.0 | 0.0 |
| Soybean Meal | 0.0 | 0.0 | 300.0 |
| Gelatin | 50.0 | 35.0 | 35.0 |
| Copper Supplement | 1.0 | 0.7 | 0.7 |
| Artemia Cysts | 25.0 | 17.5 | 17.5 |
| Calcium Phosphate | 16.5 | 11.6 | 11.6 |
| Chromic Oxide | 5.0 | 3.5 | 3.5 |
| Vitamin/Mineral Premix (Taplow) | 2.5 | 1.8 | 1.8 |
To determine the digestibility of Duckweed and soybean meal diets, adult 'feeder-sized' males (average weight: 10.7 ± 0.2 g, n = 234) were held in 18 tanks. Each tank contained 13 crayfish, each housed in separate mesh-bottom chambers. Crayfish were acclimated to the same experimental diets (i.e., REF, DW, SBM) for a period of six weeks prior to the beginning of faecal collection. Faecal collection was carried out over a period of ten days. Samples from two tanks, for each diet treatment, were randomly selected and pooled in order to collect enough sample for analysis. Therefore each sample contained the faeces produced by 26 individuals over ten days. An estimate of faecal production for each tank, standardized to biomass, was generated. Chromic oxide was included in each diet as an inert marker in order to calculate apparent digestibility (dry matter and protein) of each diet and substitute ingredient.
Results
Broodstock fertilized in captivity produced an average of 58 ± 7 embryos (mean ± SE) per individual. Beginning on April 22, 2011, stage I juveniles began to moult to stage II over a period of one week. Approximately 1,500 stage II juveniles were collected in total. Each of the diets tested showed very good survival using the vertical incubator set-up. Survival was 90, 90, and 81 % for the REF, DW, and SBM diets, respectively, for a 365 day period (Fig. 2). Although it was difficult to determine the exact number of moults for each individual, due to the crayfish occasionally consuming their own exuvia, the maximum number of moults observed was 13.
Crayfish fed the DW diet showed increased pigmentation compared to the REF and SBM diets. Within each treatment, a large amount of variation in carapace length and mass was observed and there was little difference in either average carapace length or mass between the three dietary treatments (Fig. 3).
The maximum and minimum carapace lengths observed for fed animals at the end of the study were 27.0 and 9.8 mm, respectively. The maximum and minimum masses were 5.0 and 0.2 g, respectively.
Figure two shows the average survival rate of juvenile crayfish over time when fed three test diets. The horizontal axis shown at bottom measures time from 0 to 400 days. The vertical axis at left measures the percentage of surviving crayfish, ranging from 0 at the bottom to 100 percent at the top. The approximate average survival rate of crayfish fed the reference diet (dark blue line) and the Duckweed diet (red line) was approximately 90% after 365 days. The average survival rate of crayfish fed the soybean meal diet was approximately 85% after 365 days. The light blue line, low on the chart, indicates the average survival rate of an unfed control group of crayfish was approximately 15% after 365 days.
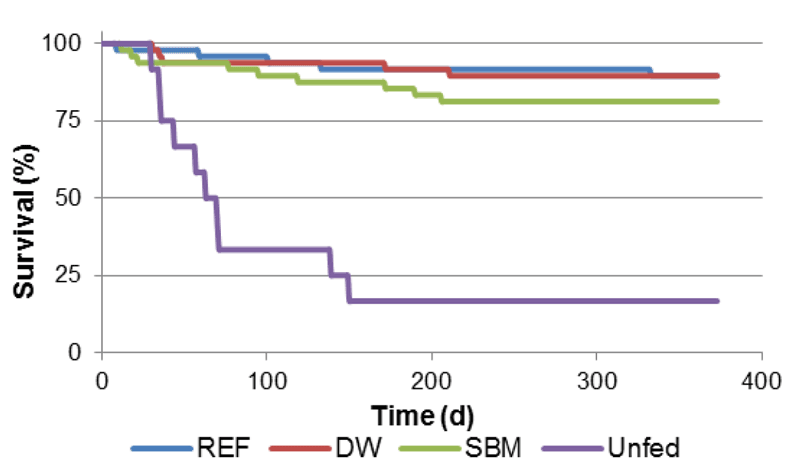
Figure 2
Survival of juvenile signal crayfish exposed to three different diet treatments and a control (unfed).Figure 3A depicts the average length to which crayfish carapace (bodies) grew over 365 days when exposed to three test diets. The bottom horizontal axis measures time in days and the vertical axis measures length from 0 at the bottom to 20 millimeters (mm) at the top. Crayfish fed the reference diet (dark blue line,) the Duckweed diet (red line) and the soybean meal diet (green line) averaged a length of approximately 15 mm after 365 days. The unfed control group of crayfish (light blue line) were approximately 5 mm in length after 365 days. Figure 3B shows the average mass (weight) of crayfish exposed to three test diets, with the horizontal axis at bottom showing time in days and the vertical axis at left showing body mass in grams, from 0 to 2. Crayfish fed the reference diet (dark blue line,) the Duckweed diet (red line) or the soybean meal diet (green line) measured an average weight of approximately 2 grams after 365 days. Crayfish in an unfed control group (light blue line) measured an average weight of approximately 0.25 grams after 365 days.

Figure 3
A) average Carapace length and B) average mass of juvenile signal crayfish exposed to three different diet treatments and a control (unfed). Values are mean (n = 42).Adult crayfish showed similar patterns in their overall apparent dry matter and protein digestibility for each diet, as well as their apparent dry matter and protein digestibility for each substitute ingredient (Fig. 4). The REF and SBM diets had similar digestibility (84 and 85 %, respectively), whereas the digestibility of the DW diet was about 15 % lower (70 %). However, over the same collection period, animals fed the Duckweed diet produced over twice as much faeces (i.e., 1.1 mg dry mass g-1 crayfish d-1 against 0.4 and 0.5 for REF and SBM diets respectively). Although the digestibility of Duckweed was noted to be lower, when faecal production and digestibility were used to estimate feed intake, crayfish consumed about 20 % and 50 % more of the DW diet than the SBM and the REF diet, respectively. This suggests that aquatic macrophytes may be important feed ingredients for increasing the palatability and therefore caloric intake, regardless of digestibility.
Figure 4 is a bar graph that measures the average digestibility of three crayfish test diets according to the dietary dry matter and crude protein content of the formulated diets, and separately measures the digestibility of the dry matter and crude protein in Duckweed and soybean meal. The vertical axis shows digestibility measured from 25% at the bottom to 100% at the top. There are four groups of bars, organized from left to right. Dark blue bars indicate the reference diet. The red bars indicate the Duckweed diet and green bars indicate the soybean meal diet. At far left, the first group of three bars shows the percentage digestibility of the apparent dry matter (ADMD) in the test diets. The ADMD of the reference diet (blue bar) and the soybean meal diet (green bar) measured approximately 80% digestible and the Duckweed diet (red bar, centre) measured approximately 70% digestible. The second group of three bars shows the digestibility of apparent crude protein (ACPD) in each diet. The ACPD in the reference diet (blue bar) and the Soybean Meal diet (green bar) measured approximately 80% digestible. The ACPD in the Duckweed diet (red bar, centre) measured approximately 75% digestible. The third group of two bars shows the digestibility of the apparent dry matter in the two main ingredients. Apparent dry matter in Duckweed (red bar) measured approximately 35% digestible and dry matter in soybean meal (green bar) measured approximately 80% digestible. The fourth and last group of two bars at far right shows the digestibility of the apparent crude protein in the two main ingredients. Apparent crude protein in Duckweed (red bar) measured approximately 65% digestible and crude protein in soybean meal (green bar) measured approximately 95% digestible.
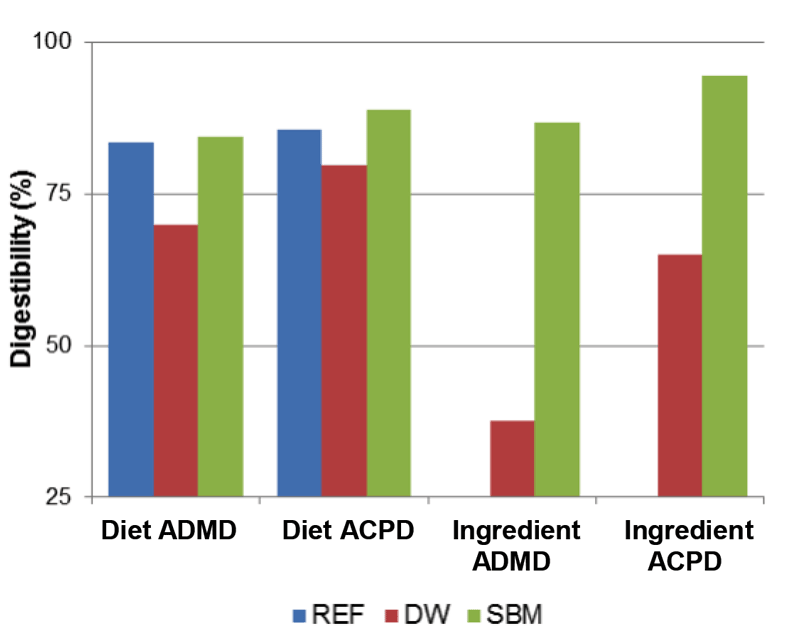
Figure 4
Apparent dry matter (ADMD) and crude protein (ACPD) digestibility for each compound diet and for the ingredients (duckweed and soybean meal) separately. Values are mean (n = 3).Conclusions
This project was successful in demonstrating new culture techniques in the production of Signal Crayfish in British Columbia. During this project, individuals hatched from wild broodstock, fertilized in the laboratory, were successfully raised for an entire year with minimal mortalities. The system of vertical incubators used for hatching and rearing juvenile crayfish provided an effective means of intensively producing juveniles that could be used for outplanting to culture ponds for grow-out. Three different artificial-feed formulations were developed for Signal Crayfish and shown to provide good growth rates. Results suggest that aquatic macrophytes may be important feed ingredients for increasing palatability and caloric intake. Co-culturing aquatic macrophytes in semi-intensive or extensive crayfish culture ponds is likely a cost-effective and sustainable means of feeding crayfish.
A key next step in the development of Signal Crayfish culture in Canada will be to extend the findings of this study to larger-scale culture operations. This can be achieved through the production of larger numbers of juvenile crayfish in hatchery operations and seeding them into semi-intensive or extensive culture ponds which have been supplemented with aquatic macrophytes and complex substrates. The current study used diets with protein, carbohydrate, and lipid contents that fell within the range reported to produce optimal growth. Further studies refining these ratios and examining how different sources for these nutrients affect growth will be helpful in optimizing intensive culture techniques. In addition, as little difference in growth and survival was found between juvenile Signal Crayfish fed different diets, but increased caloric intake for adult crayfish fed the Duckweed diet, studies investigating the nutritional needs of different life stages may be of benefit.
This ACRDP project (P-10-01-001) was a collaborative effort among the Department of Fisheries and Oceans (DFO Science) and Asturia Aquaculture Crayfish Consulting and Broken Briar Enterprises Ltd. This research was carried out by Dr. Dan Curtis ( Dan.Curtis@dfo-mpo.gc.ca), supported by the Visiting Fellowship in Canadian Governmental Laboratories program of the Natural Sciences and Engineering Research Council of Canada. Research project lead, Dr. Chris Pearce can be contacted at Chris.Pearce@dfo-mpo.gc.ca.
For further information on this and other ACRDP projects, visit: Aquaculture Collaborative Research and Development Program (ACRDP)
Published by:
Aquaculture Science Branch,
Fisheries and Oceans Canada
Ottawa, Ontario K1A 0E6
©Her Majesty the Queen in Right of Canada 2009
ISSN 1919-6806 (Print)
ISSN 1919-6814 (Online)
ISBN 978-1-100-21754-3
DFO/2013-1854
Cat No. Fs48-2/17-2013E-PDF
French version and alternative formats available through: Programme coopératif de recherche et développement en aquaculture (PCRDA)
- Date modified: