Geoduck clam (Panopea generosa): Anatomy, Histology, Development, Pathology, Parasites and Symbionts
Intracellular Prokaryotes of Geoduck Clams
Clusters (microcolonies) of intracellular prokaryotes believed to be species of Rickettsea or Chlamydia were observed in epithelial cells of the gill filaments (Figs. 1a and 1b) and occasionally in the epithelium of the palps. Because of their appearance they are often referred to as basophilic inclusion bodies. Although the prevalence of infection in geoduck clams is high (almost all adult clams infected), the intensity of infection is usually very low which has hindered the specific identification of these parasites.
Figures 1a and b. Inclusion bodies (arrows) in histological sections of gill epithelial cells with no indication of an adverse response by the geoduck clam. Hematoxylin and eosin stain.
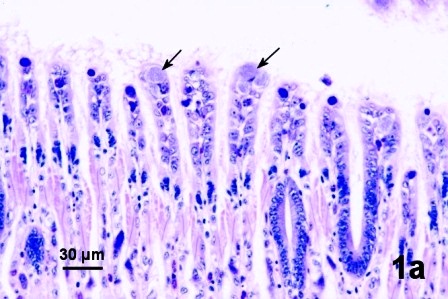
Figure 1a. Inclusion bodies (arrows) in a histological section of gill epithelial cells with no indication of an adverse response by the geoduck clam.

Figure 1b. Inclusion bodies (arrow) in histological sections of gill epithelial cells with no indication of an adverse response by the geoduck clam.
Inclusion bodies have been observed in other bivalves of British Columbia and the causative agent(s) are not known to be pathogenic in most cases. Dorfmeier et al. (2015) also reported inclusion bodies (intracytoplasmic Rickettsia-like colonies) in the gill epithelial cells of P. generosa from Washington State. “Rickettsia-like” inclusions were only detected inside the epithelial cells of the secondary digestive diverticula and not the gills (ctenidial epithelial cells) of a few P. generosa surveyed from the Pacific coast of Baja California, Mexico (Cáceres-Martínez et al. 2015). In all cases there was no evidence of significant pathology associated with infection.
Intracellular prokaryotes belonging to the Rickettsiales have also been reported in the gills and digestive gland epithelial cells of many species of clam around the world including Cerastoderma edule, Donax trunculus, Mercenaria mercenaria, Mya arenaria, Venerupis (=Tapes) philippinarum, Tapes (=Ruditapes) decussatus, Siliqua patula, Tapes pullastra, Tellina tenuis, Tridacna crocea and Hippopus hippopus as well as a wide variety of marine molluscs including oysters, mussels and scallops. Infections are usually light and usually not associated with disease. However, extensive inflammation of gill tissue and mortalities in cultured giant clams (H. Hippopus) were associated with heavy gill infections of Rickettsiales-like organisms in the Philippines and Micronesia (Norton et al. 1993). Also, a similar organism, 'Candidatus Xenohaliotis californiensis', that inhabits cells of the digestive gland of abalone has been accused of causing foot withering syndrome and mass mortalities of black abalone in California (Gardner et al. 1995, Friedman et al. 2000).
Methods of control
No known methods of prevention or control. It was suggested that overcrowding and low exchange rates of water in land based culture tanks predisposed Hippopus hippopus to increased intensity of infection, clinical disease, and mortalities. Experiments with 'Candidatus Xenohaliotis californiensis' indicated that this pathogen could be transmitted via the water column and did not require direct contact between infected and uninfected abalone and above normal temperatures seem to have a synergistic effect on the disease (Martínez et al. 2000, Moore et al. 2000).
References
Cáceres-Martínez, J., R. Vásquez-Yeomans and R. Cruz-Flores. 2015. First Description of symbionts, parasites, and diseases of the Pacific geoduck Panopea generosa from the Pacific coast of Baja California, Mexico. Journal of Shellfish Research 34: 751-756.
Comps, M. and R. Raimbault. 1978. Infection rickettsienne de la glande digestive de Donax trunculus Linné. Sciences et Pêches, Bulletin de l'Institute des Pêches Maritimes 281: 11-12. (In French).
Comps, M. 1982. Étude morphologique d'une infection rickettsienne de la palourde Ruditapes philippinarum Adam and Reeves. Revue des Travaux de l'Institut des Pêches Maritimes 46: 141145. (In French).
Comps, M. 1983. Infections rickettsiennes chez les mollusques bivalves des côtes francaise. Rapports et Procèsverbaux des Réunions du Conseil International pour l'Exploration de la Mer 182: 134136. (In French).
Dorfmeier, E.M., B. Vadopalas, P. Frelier and C.S. Friedman. 2015. Temporal and spatial variability of native geoduck (Panopea generosa) endosymbionts in the Pacific Northwest. Journal of Shellfish Research 34: 81-90.
Elston, R.A. 1986. Occurrence of branchial rickettsiales-like infection in two bivalve molluscs, Tapes japonica and Patinopecten yessoensis, with comments on their significance. Journal of Fish Diseases 9: 6971.
Friedman, C.S., K.B. Andree, K.A. Beauchamp, J.D. Moore, T.T. Robbins, J.D. Shields, and R.P. Hedrick. 2000. 'Candidatus Xenohaliotis californiensis', a newly described pathogen of abalone, Haliotis spp., along the west coast of North America. International Journal of Systematic Evolution and Microbiology 50: 487-855.
Gardner, G.R., J.C. Harshbarger, J.L. Lake, T.K. Sawyer, K.L. Price, M.D. Stephenson, P.L. Haaker and H.A. Togstad. 1995. Association of prokaryotes with symptomatic appearance of withering syndrome in black abalone Haliotis cracherodii. Journal of Invertebrate Pathology 66: 111-120.
Harshbarger, J.C., S.C. Chang and S.V. Otto. 1977. Chlamydiae (with phages), mycoplasmas, and rickettsia in Chesapeake Bay bivalves. Science 196: 666668.
Martínez, J.C., C.A. Tinajero, Y.G. Rentería and J.G.G. Avilés. 2000. Rickettsiales-like prokaryotes in cultured and natural populations of the red abalone Haliotis rufescens, blue abalone, Haliotis fulgens, and the yellow abalone Haliotis corrugata from the Baja California, Mexico. (Abstract) Journal of Shellfish Research 19:503.
Meyers, T.R. 1979. Preliminary studies on a chlamydial agent in the digestive diverticular epithelium of hard clams Mercenaria mercenaria (L.) from Great South Bay, New York. Journal of Fish Diseases 2: 179189.
Moore, J.D., T.T. Robbins and C.S. Friedman. 2000. The role of a Rickettsia-like prokaryote in withering syndrome in California red abalone, Haliotis rufescens. (Abstract) Journal of Shellfish Research 19: 525-526.
Norton, J.H., M.A. Shepherd, M.R. Abdon-Naguit and S. Lindsay. 1993. Mortalities in the giant clam Hippopus hippopus associated with rickettsiales-like organisms. Journal of Invertebrate Pathology 62: 207-209.
Citation Information
Bower, S.M. and Blackbourn, J. (2003): Geoduck clam (Panopea generosa): Anatomy, Histology, Development, Pathology, Parasites and Symbionts: Intracellular Prokaryotes of Geoduck Clams.
Date last revised: August 2020
Comments to: Susan Bower
- Date modified: