Canadian Aquaculture R&D Review 2017
Fish Health
Study of Genomic Diversity in Aeromonas salmonicida, the Etiological Agent that Causes Furunculosis, to Establish its Resistome, Epidemiological Markers, and Potential Treatments
The Aeromonas salmonicida bacterium is the infectious agent that causes furunculosis in salmonids (salmon, trout, Arctic Charr, etc.). Controlling this disease, which is very harmful to the aquaculture industry, can prove to be quite demanding and fruitless, mainly due to the logistic constraints of the vaccination and the very frequent resistance of A. salmonicida to several antibiotics. We are therefore studying the genomic diversity of A. salmonicida to better understand its virulence and its antibiotic resistome (all genes conferring resistance to antibiotics). Through familiarization with this diversity, tools and alternative treatments for preventing or curing furunculosis can be created. In concrete terms, we are developing a kit for quickly diagnosing antibiotic resistance. We are also studying the action of mobile DNA elements in the evolution, host adaptation, and geographic distribution of A. salmonicida, and their potential activation by various treatments, including the effect of certain essential oils, with a view to developing a treatment. It is also our intent to verify the potential of bacteriophages (viruses that infect bacteria) as a cure for furunculosis. It is a recurrent disease, hard to control and, as such, all of these approaches must be considered.
Increased knowledge of the pathogen, A. salmonicida, will aid Canadian aquaculture productivity while enabling fish farmers to: 1) make appropriate use of antibiotics during treatment; 2) better track the pathogen’s location; and 3) suggest alternatives to antibiotics as treatment. Ultimately, our work will offer an integrated approach to follow, control, and treat A. salmonicida infections.
Date: NOV. 2012–MAR. 2019
Funded by: Natural Sciences and Engineering Research Council (NSERC)
Co-Funded by: Ressources Aquatiques Québec (RAQ); Ministère de l’Agriculture, des Pêcheries et de l’Alimentation du Québec (MAPAQ)
Project Lead: Steve Charette (U Laval)
Project Team: Antony Vincent, Valérie Paquet, Katherine Tanaka, Sabrina Attéré, Alex Bernatchez, Mélanie Trudel, Jean-Guillaume Edmond-Rheault (U Laval)
Collaborators: Nicolas Derome, Michel Frenette, Sylvain Moineau (U Laval); Andrée Lafaille (UdeM)
Contact: steve.charette@bcm.ulaval.ca
Website: www.amibe.org
The Effect of Dietary Camelina Oil on the Health of Salmon
Traditional salmon feeds use high levels of fishmeal and fish oil to meet the nutritional needs of the fish, but these ingredients face large fluctuations in price and availability. Lower-cost alternatives have been investigated, including canola oil, soy oil, and poultry fat. Oil from the plant species Camelina sativa is another promising option that is already able to replace fish oil in diets for Atlantic Cod, Atlantic Salmon, and Rainbow Trout with little or no reduction in growth or feed efficiency. However, substitution of dietary fish oil with vegetable oils (such as Camelina oil) in fish feed may elicit inflammatory changes due to the different fatty acid profiles. There are limited data available on changes to the gut, and no data for vegetable oil’s health effects on Chinook Salmon.
This project seeks to examine the influence of fish oil substitution by Camelina oil on susceptibility to intestinal inflammation of Chinook Salmon under culture conditions. Inflammation is energetically demanding and stressful, thus influencing disease resistance. By using Camelina oil to trigger gut inflammation in Chinook Salmon, results from this project will increase knowledge and understanding of these effects in order to develop better management practices surrounding disease impacts on cultured finfish species.
Start Date: SEP. 2015–OCT. 2016
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-funded by: Creative Salmon Co. Ltd.
Project Lead: Ian Forster (DFO)
Project Team: Simon Jones (DFO); Marije Booman (UVic); Barb Cannon, Tim Rundle (Creative Salmon Co. Ltd.); Jack Grushcow (Linnaeus Plant Sciences Inc.); Brad Hicks (Taplow Ventures, Ltd.)
Collaborators: Barb Cannon, Tim Rundle (Creative Salmon Co. Ltd.)
Contact: Ian.Forster@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/15-2-P-03-eng.html
Prophylactic Effect of Haslea ostrearia Culture Supernatant Containing the Pigment Marennine Against the Pathogenic Bacteria Vibrio splendidus in Bivalve Hatcheries
A primary requisite in any shellfish culture or farming operation is an abundant, reliable, and inexpensive supply of juveniles. Hatchery-produced seed is increasingly becoming the standard raw material for aquaculture. However, repeated bacterial infections result in heavy mortalities, causing major losses and great expenses for shellfish growers. This project explores the possibility of using the supernatant of cultured diatoms (Haslea ostrearia) containing marennine, a natural microalgal pigment, as an antimicrobial agent in bivalve hatcheries. The Blue Mussel (Mytilus edulis) and the Sea Scallop (Placopecten magellanicus) were used as model animals, and the pathogenic marine bacteria, Vibrio splendidus, was used to induce larval mortality. The hypothesis tested was that V. splendidus pathogenicity in larval rearings can be controlled by using marennine-containing culture supernatants. The effect of three marennine concentrations was tested on a larval rearing over 20 days for M. edulis and 9 days for P. magellanicus. At a low dose (0.1 mg/L), survival and physiological condition were both higher than in the control. In bacterial challenges, larvae were exposed to V. splendidus for 72 hours, with or without marennine. The bacterial challenge caused significant mortality when compared to control, while the marennine-treated larvae showed a significantly higher survival. The results show that marennine is an interesting molecule for pathogen control in hatcheries as it is active at low concentrations and can significantly enhanced larval survival and physiological condition.
Date: MAY 2013–FEB. 2016
Funded by: Natural Sciences and Engineering Research Council (NSERC)
Co-Funded by: Ressources Aquatiques Québec (RAQ); Fermes Marines Inc.
Project Lead: Réjean Tremblay (UQAR–ISMER)
Project Team: François Turcotte, Bertrand Genard, Karine Lemarchand, Réjean Tremblay (UQAR–ISMER); Jean-Luc Mouget (U Maine); Jean-Sébastien Deschênes (UQAR)
Contact: Rejean_Tremblay@uqar.ca
Investigating Probiotic Bacteria and Their Bacteriocins as Part of a Disease Management Strategy in Salmon Aquaculture
On salmon farms, diseases caused by bacterial infections are managed by minimizing the risk of exposure to pathogens, vaccination, and ensuring the availability of sufficient high quality water and feed. There are concerns regarding the effectiveness and long term sustainability of using antibiotics to treat bacteria, so to help further reduce antibiotic use, this study focused on the therapeutic potential of probiotics in aquaculture.
Probiotic bacteria can enhance the immune system of the fish, and can change the composition of the intestinal microbiota, which function in overall fish health. Probiotic bacteria may also secrete antimicrobial compounds that kill other bacteria, including fish pathogens. This project used probiotic bacteria and antimicrobials with activity against common salmon pathogens, and evaluated their application as feed additives to improve fish health.
This project supports the DFO objective of optimal fish health.
Specifically, this project found the following results:
- Paenibacillus terrae and Paenibacillus polymyxa showed antimicrobial activity against five salmon pathogens. P. terrae was selected for further trials as it showed a stronger activity.
- There was no evidence that the probiotic diets caused a reduction in feed attraction or appetite.
- No significant effect of the probiotic diets was found on the survival rate of salmon after exposure to the pathogen Vibrio anguillarum.
- P. terrae survived in salmon intestines but did not colonize the gut.
- The results suggested that the live P. terrae diet may cause changes in the abundance and/or the diversity of the intestinal microbiota. This is currently being examined in more detail.
- Increased knowledge of the effect of P. terrae on fish health will help determine its suitability as a therapeutic probiotic feed additive for salmon, which will benefit the aquaculture industry.
Date: APR. 2014–JUN. 2015
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Mainstream Canada (Cermaq Canada Ltd.); Marine Harvest Canada Limited
Project Lead: Simon Jones (DFO)
Project Team: Marije Booman, Steve Cho (DFO)
Collaborators: Peter McKenzie (Mainstream Canada (Cermaq Canada Ltd.)); Diane Morrison (Marine Harvest Canada Limited)
Contact: Simon.Jones@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/P-14-01-003-eng.html
The Effects of Smolt Size on the Intensity of Kudoa thyrsites Infections in Atlantic Salmon
Atlantic Salmon infected with Kudoa thyrsites do not exhibit clinical signs of disease. However, protease secretion from this parasite rapidly deteriorates affected muscle in the salmon when the fillet is processed, resulting in economic loss for the grower. Earlier research suggested the risk of K. thyrsites was reduced when salmon were transferred to sea as larger smolts. This research project involves a more robust test of the size hypothesis by conducting trials that examine how smolts of a range of size classes respond to exposure to the K. thyrsites parasite.
The results of this project may help to improve the competitiveness of the Canadian salmon aquaculture industry by assessing a problem that greatly affects the marketability of Atlantic Salmon produced in British Columbia. An understanding will be gained in the relative importance of fish size (smolts) in influencing the development of K. thyrsites. Data obtained from the project will help inform K. thyrsites management strategies.
Start Date: OCT. 2015–JUN. 2017
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-funded by: Marine Harvest Canada Limited; Cermaq Canada Ltd.
Project Lead: Simon Jones (DFO)
Collaborators: Diane Morrison (Marine Harvest Canada Limited); Kathleen Frisch (Cermaq Canada Ltd.)
Contact: Simon.Jones@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/15-2-P-01-eng.html
Comparison of Field Isolates of Moritella viscosa: Characterization and in Vivo Challenge Model Development to Address Winter Ulcer Mitigation in Canada
The bacteria Moritella viscosa is associated with cold-water ulcer disease in farmed Atlantic Salmon at sea. Over the past decade, skin ulcers have also been noted in warmer water in Eastern Canada. The role of M. viscosa remains unclear due to its infrequent recovery from summer ulcers, however, suspicion of its involvement persists. At the onset of this research, there was no vaccine for ulcer disease based on Canadian isolates. Salmon producers had limited tools to manage this disease, and though commercial vaccines used in Europe existed, no information existed with regard to their efficacy against Canadian isolates.
For this project, Canadian field isolates were collected and characterized by establishing in vitro growth curves at different temperatures. Differences in protein profiles and immunogenic proteins through gel electrophoresis and Western blotting methods were examined. Ultimately, the characterization steps allowed for the development of an in vivo challenge model using an isolate of adequate virulence with appropriate culturing and challenge exposure methodologies. When employed at 8°C, the model was successful in inducing lesions consistent with clinical cold-water sores found on sea cage Atlantic Salmon. In addition, the live challenge model also verified that a recent Atlantic isolate, originating from a summer ulcer field case, was virulent at 12°C and caused lesions in unvaccinated animals at this warmer temperature. Diagnostically, M. viscosa can be recovered by bacteriology with a high success rate from all stages of lesions present on challenged fish. The developed challenge model can be used for evaluating and developing potential commercial vaccines and was shown to be an excellent model for the study of ulcerative disease progression.
Date: AUG. 2014–MAR. 2016
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Cooke Aquaculture Inc.; Novartis Animal Health Canada Inc.
Project Lead: Steven Leadbeater (DFO)
Project Team: Anthony Manning (RPC); Leighanne Hawkins (Cooke Aquaculture Inc.); Allison MacKinnon (Novartis Animal Health Canada Inc.)
Contact: Steven.Leadbeater@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/M-14-01-003-eng.html
Estimating the Potential for Waterborne Transmission of Infectious Haematopoietic Necrosis (IHN) Disease between Salmon Farms and Wild Sockeye Salmon in the Discovery Islands, British Columbia
Infectious Haematopoietic Necrosis Virus (IHNV) is responsible for major economic losses in British Columbia salmon aquaculture operations. Viral transmission patterns and oceanographic circulation models for the Discovery Islands region provide estimates for the potential of IHNV outbreak and spread to neighbouring farms.
This study quantified the transmission potential of IHN disease between farms and wild Sockeye Salmon. It also determined the capacity of vaccinated Atlantic Salmon and IHNV-carrying Sockeye Salmon to transmit the virus to naïve hosts (ones who have never before been infected).
Specifically this project found the following results:
- Neighbouring naïve farms can become infected via waterborne transmissions. Adherence to the stringent disease management practices (such as immediately quarantining an infected farm site) reduces transmission of infectious doses to other farms.
- The model simulations demonstrated that using commercially available IHN vaccines greatly reduces the risk of transmission by lowering the overall number of susceptible fish. It also reduced the virus shedding capacity of the vaccinated farm population if it were to become infected with IHNV. To date, vaccinated farmed Atlantic Salmon populations have not developed IHN disease, while unvaccinated populations have been declared INHV positive.
- With some modifications, the modelling tools can be applied to other diseases and to simulate transmission from farmed to wild populations and vice versa. It will be possible to estimate the risk of infection for fish swimming in proximity to diseased farms and assess the relative effectiveness of disease and farm management practices such as vaccination, quarantine, depopulation, and determining optimal locations for aquaculture sites.
Date: JUN. 2013–JUN. 2015
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Marine Harvest Canada Limited; Grieg Seafood BC Ltd.; Mainstream Canada (Cermaq Canada Ltd.)
Project Lead: Kyle Garver (DFO)
Project Team: Mike Foreman, Darren Tuele, Ming Guo, Dario Stucchi, Peter Chandler (DFO)
Collaborators: Diane Morrison (Marine Harvest Canada Limited); Barry Milligan (Grieg Seafood BC Ltd.); Peter McKenzie (Mainstream Canada (Cermaq Canada Ltd.))
Contact: Kyle.Garver@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/P-13-01-001-eng.html

Aerial photo of the Discovery Islands in British Columbia. Photo: Kyle Garver (DFO)
Optimizing the Fish Health Reporting and Data Management System
The Centre for Coastal Health (CCH) is conducting a utility assessment of the current BC Salmon Farmers Association (BCSFA) fish health database, which houses fish health data contributed by industry members from 2001-2016. The results of the utility assessment will be used to determine how the database can be used to generate useful fish health information for BCSFA members and other stakeholders, including the public. The CCH is also developing a portfolio of fish health reports from data contained within the database.
The overall objectives of this project are to: 1) understand fish health information and accessibility needs, and to determine the uses and limitations of the BCSFA fish health database; 2) design new fish health reports from the BCSFA database that assist farm management and aid in communications; and 3) identify changes that could be made to the database to expand its utility.
The current fish health database functions as a repository of data, from which limited analysis can be conducted. The outcomes from this project should improve the functionality of the database, and enable improved reporting of fish health events.
Date: APR. 2015–MAR. 2016
Funded by: BC Salmon Farmers Association
Project Lead: Carl Ribble (CCH)
Project Team: Tyler Stitt, Theresa Burns, Stefan Iwasawa (CCH)
Collaborators: Kyle Garver (DFO)
Contact: carl.ribble@gmail.com
Website: www.centreforcoastalhealth.ca
Screening of Cultured Atlantic Salmon for Resistance and Susceptibility to Infection by Sea Lice (Lepeoptheirus salmonis) and Renibacterium salmoninarum, the Causative Agent of Bacterial Kidney Disease (BKD)
While vaccination is one approach to impart disease resistance, there is significant variation among Atlantic Salmon in terms of vaccine responsiveness. It is imperative to assess the genetic stocks of salmon for their robustness against disease.
This research project will test family crosses of farmed salmon for resistance to Lepeoptheirus salmonis (sea lice) and Renibacterium salmoninarum (the causative agent of Bacterial Kidney Disease, BKD). Testing of more than one disease agent (in this case, L. salmonis and R. salmoninarum) will help assess what resistance to one agent type may mean for resistance to another common agent. Information resulting from this project will aid in broodstock selection with the goal of enhancing resistance of farmed fish to both sea lice and BKD infections. This will serve to reduce losses to farmed fish and improve the sustainability of the Canadian aquaculture industry.
Date: SEP. 2015–JUN. 2018
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Kelly Cove Salmon Ltd. (KCS)
Project Lead: Steven Leadbeater (DFO)
Project Team: Keng Pee Ang (KCS); Jake Elliott, Frank Powell (Cooke Aquaculture Inc.); Anthony Manning (RPC)
Collaborators: Keng Pee Ang (KCS)
Contact: Steven.Leadbeater@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/15-1-M-01-eng.html

Post mortem examination of salmon from Renibacterium salmoninarum exposure challenge. From foreground Melissa Holborn (U Guelph), Steven Leadbeater, and Ann Kinney (DFO). Photo: Cindy Hawthorne (DFO)

Project team members collecting gravid female sea lice on a harvest boat. Egg strings will be hatched to be used in exposure challenges. Clockwise from top left, Cindy Hawthorne, Steven Leadbeater (DFO), and Melissa Holborn (U Guelph). Photo: DFO

Cindy Hawthorne (DFO) collecting samples from fish following a sea lice exposure challenge. Photo: Melissa Holborn (U Guelph)
Microbial Impacts on Shellfish Aquaculture in Relationship to Ocean Acidification
Ocean acidification (characterized by high dissolved carbon dioxide (pCO2) levels) can affect shellfish fertilization rates, hatching success, larval development, shell deformation, seed production, and survival. However, it is not clear whether high pCO2 levels alone are responsible for poor performance in some West Coast shellfish operations. While hatcheries have successfully controlled levels of pCO2 for larval and nursery production through the use of buffering systems, there have continued to be high variations in batch to batch survival. This suggests that other uncontrolled aspects of the seawater composition during high pCO2 events may be at play, most notably bacterial compositions. Researchers are applying a metabarcoding approach to assess the shifts in bacterial community composition that occur in sea water coming into a Pacific Northwest shellfish hatchery, post pCO2 buffering, and throughout larval culture and comparing bacterial compositions in larvae and water between batches of good and poor performance. Ultimately, the project will identify the degree to which initial pCO2 levels impact bacterial communities of cultured shellfish and identify whether certain bacterial species are more common in seed batches demonstrating poor survival.
The knowledge obtained through this research will be useful in the development of a mitigation strategy to reduce the impact of ocean acidification on the shellfish industry on Canada’s West Coast. Additionally, the identification of factors that contribute to farmed shellfish mortality may lead to the development of monitoring tools. This would ultimately assist the industry in enhancing environmental sustainability and economic viability of shellfish aquaculture.
This project supports the DFO objective of optimal fish health.
Date: JUN. 2014–JUN. 2017
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Island Scallops Ltd.; Fanny Bay Oysters Ltd.
Project Lead: Kristi Miller-Saunders (DFO)
Collaborators: Barb Bunting, Rob Saunders (Island Scallops Ltd.); Brian Yip (Fanny Bay Oysters Ltd.)
Contact: Kristi.Saunders@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/P-14-02-001-eng.html
Low Pathogenic Infectious Salmon Anemia Virus (ISAV) in Vivo: A Comparative Genomic Study
Infectious Salmon Anaemia (ISA) is a threat to the salmon aquaculture industry. This study investigated different Infectious Salmon Anemia Virus (ISAV) strains with varying degrees of virulence (harmful effects). The virus was observed in vivo in Atlantic Salmon and the infectious potential of surviving/carrier fish was evaluated. Tested fish showed significant differences in susceptibility. Specifically, this project found the following results:
- Fish exposed to a low pathogenic ISAV had detectable viral RNA in their tissues 18 months past exposure.
- Fish surviving a high pathogenic ISAV returned to a near negative state rapidly after the end of mortalities, but are also potential long term carriers of ISAV.
- Low pathogenic ISAV can be transmitted horizontally and create a state similar to “herd immunity”, where a proportion of fish were exposed and are thus naturally immune to ISAV infections, thus reducing the risk of an outbreak caused by a virulent ISAV. Still, those fish could continue to transmit infections due to their long term carrier state.
- Low virulence ISAV strains showed little to no fish mortalities, thus confirming observations in the field about their low pathogenicity.
- The high virulence ISAV strains produced between ~40% and 80% mortalities. The differences in observed mortality using this strain shows the possibility of selecting fish with specific genotypes that might be associated with favourable traits such as disease resistance, which is beneficial for breeding programs and the aquaculture industry.
Date: DEC. 2011–JUN. 2016
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Kelly Cove Salmon Ltd. (KCS)
Project Lead: Nellie Gagné (DFO)
Project Team: Francis Leblanc, Steven Leadbeater, Mélanie Roy, Mark LaFlamme (DFO)
Collaborators: Keng Pee Ang (KCS)
Contact: Nellie.Gagne@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/MG-11-01-004-eng.html

Necropsy of Atlantic Salmon to detect the presence of Infectious Salmon Anemia. Photo: (DFO)

Atlantic Salmon infected with Infectious Salmon Anemia–operculum removed to show pale gills. Photo: Nellie Gagné (DFO)
The Effects of Prior Exposure and Body Size on the Intensity of Kudoa thyrsites Infections in Farmed Atlantic Salmon
The parasite Kudoa thyrsites is the cause of soft-flesh syndrome in post-harvest Atlantic Salmon farmed in British Columbia. Infected fish exhibit no clinical signs of disease, but affected muscle deteriorates after processing depending on the severity of infection. The industry in BC is subject to substantial economic loss because of Kudoa-associated changes to fillet quality. Currently, there are no vaccines or approved strategies for treatment for K. thyrsites.
This study investigated three management options for K. thyrsites infections:
- The efficacy of ultraviolet irradiation of seawater as a method to inactivate K. thrysites in the laboratory.
- The influence of prior exposure to K. thyrsites on parasite development during subsequent exposure.
- The influence of fish size on the prevalence and severity of the infection.
The knowledge gained through this research project will lead to a greater understanding of disease transmission and wild-farmed interactions. Additionally, this information will allow for better finfish cage siting and decision making and the development of tools to help further manage this disease.
This project supports the DFO objective of optimal fish health.
Specifically, the following results were found:
- Ultraviolet irradiation of seawater significantly reduces risk of infection in Atlantic Salmon. UV-irradiation is not practical in current net pen production systems, but would prove valuable in a closed-containment system.
- Previous infection with K. thyrsites reduces the risk of a severe subsequent infection. This is consistent with observations made by industry that initial rearing of smolts at a low-risk site reduces the severity of infection following transfer to high-risk sites. It would be valuable to further investigate alternative methods of simulating an earlier exposure.
- No consistent relationship between fish size and susceptibility to K. thrysites was observed. Further research will explore alternative methods of testing the size hypothesis.
Date: JUN. 2013–JUN. 2015
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Marine Harvest Canada Limited; Mainstream Canada (Cermaq Canada Ltd.)
Project Lead: Simon Jones (DFO)
Project Team: Diane Morrison (Marine Harvest Canada Limited); Peter McKenzie (Mainstream Canada (Cermaq Canada Ltd.))
Contact: Simon.Jones@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/P-13-01-002-eng.html
Net Pen Liver Disease Project–Developing a Fluorescent Antibody Test to Prove the Causative Agent
An incidence of Net Pen Liver Disease (NPLD) was diagnosed in a population of adult Atlantic Salmon at a Grieg Seafood farm site in Nootka Sound, British Columbia, June 25, 2014. Previous presentations of the disease at company sites were limited to poor performers, three to four months post sea water entry. This new presentation was different as it affected well established grow out fish of approximately 3.6 kg. Based upon historical information, the disease had been associated with a hepatotoxic mycrocystin–LR (MC). Phase 1 of the project was to develop a fluorescent antibody test to positively identify the microcystin toxin present in affected fish.
Histological surveys of smolts (500 g) growing within an adjacent inlet did not test positive for NPLD; feed rate and mortality remained normal. Water samples were taken from the affected site and a site located geographically distant from the affected site both tested positive for the presence of MC.
Atlantic Salmon were experimentally exposed to the microcystin toxin thought to be the causative agent of the disease. Liver tissue samples were harvested over various timescales and the tissues were sent to the Animal Health Centre in Abbotsford, BC for histological processing and evaluation. Samples with lesions attributed to toxin exposure were used to develop a fluorescent antibody which was able to detect the presence of the toxin. Future work will optimize the test including determining both the level of sensitivity of the antibody and the strength of antibody required for accurate testing. This study provides proof of concept for the causative agent for Net Pen Liver Disease.
Date: AUG. 2016–MAR. 2017
Funded by: Grieg Seafood BC Ltd.
Project Lead: Tim Hewison (Grieg Seafood BC Ltd.)
Project Team: Matthew Patterson, Patrick Whittaker, Graeme Cooper (Grieg Seafood BC Ltd.)
Collaborators: Heindrich Snyman (BC Ministry of Agriculture)
Contact: Tim.Hewison@griegseafood.com
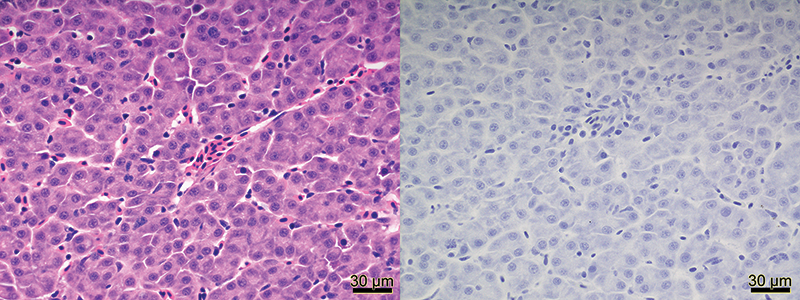
Histological pictures of normal liver tissue from an Atlantic Salmon stained by H&E (left) and stained by IHC–anti–Microcystin–LR (right). Photo: Heindrich Snyman (BC Ministry of Agriculture)

Histological pictures of liver tissue from an Atlantic Salmon exposed to the toxin Microcystin stained by H&E (left) and stained by IHC–anti-Microcystin-LR (right). Photo: Heindrich Snyman (BC Ministry of Agriculture)
Improving the Survival, Health, Quality, and Food-Safety of Post-Transported Tilapia and Barramundi
The market demand for live Tilapia (Oreochromis niloticus) and Barramundi (Lates calcarifer) in Metro-Vancouver is steadily increasing. The stress and physical injury that occurs to fish during transport from USA to Langley, B.C. for acclimation and maintenance prior to retail sales have resulted in high fish mortalities and large economic losses to Viva Aquaculture and Seafood Distribution Ltd. (Viva). Developing viable and sustainable protocols to improve the survival, health, and quality of post-transported fish will benefit the company and retailers economically, and will also provide consumers with better food safety and quality fish products. It will also lead to new economic and employment opportunities locally, as well as be a source of diversified and nutritious seafood products for consumers in British Columbia. The University of British Columbia (UBC) and Viva have established a research collaboration to improve the survival and quality of fish transported from USA to Langley, BC. The specific objectives of this collaborative project were: 1) to increase the survival rate of adult Tilapia transported from Idaho, USA, by improving water quality through the manipulation of salinity, alkalinity, hardness, pH, temperature, and dissolved oxygen, and the elimination of toxic nitrogenous wastes; and 2) to improve the quality and food safety of fish during the post-transport recuperation and acclimation period using government-approved therapeutants and prophylactics, as well as indigenous microalgae, to eliminate potential microbial pathogens that might be present in the source water used to transport the fish from USA.
The results of this research project will lead to the development of post-transport protocols that are applicable in a commercial scale; are viable, efficient, safe, and sustainable for the maintenance rearing of adult fish destined for the retail market and human consumption in Canada.
Date: FEB. 2015–DEC. 2018
Funded by: Viva Aquaculture and Seafood Distribution Ltd. (Viva)
Project Lead: Jesse Ronquillo (UBC; Viva)
Project Team: Chang Lin Ye, Kai Chen (Viva)
Collaborators: David Kitts, Siyun Wang (UBC)
Contact: jesse.ronquillo@ubc.ca
Website: www.vivaseafood.com

Tilapia (Oreochromis niloticus). Photo: Jesse Ronquillo (UBC)

Barramundi (Lates calcarifer). Photo: Jesse Ronquillo (UBC)
Piscine Reovirus (PRV): Characterisation, Atlantic Salmon Susceptibility, and Initial Survey in Farmed and Wild Salmonids in Atlantic Canada
TThe piscine reovirus or PRV is a recently identified virus that has been linked to Heart and Skeletal Muscle Inflammation (HSMI) in Atlantic Salmon. HSMI was demonstrated as an infectious disease, however, the role of PRV in HSMI continues to be studied, as PRV is often found without symptoms of HSMI. Although the host range of this virus appears primarily restricted to salmonids, it has been occasionally detected in a few non-salmonid species.
In 2015, a small sample of Atlantic Salmon from different sources on the East Coast was tested and the presence of PRV was detected for the first time. During genetic sequencing, it was discovered that the strain of virus found on the East Coast bore a close resemblance to the West Coast strain of PRV. As there are no reports of HSMI-like syndromes in salmon on the East coast, the significance of PRV detection is unclear. HSMI was recently diagnosed for the first time on the Pacific Coast; given that no cases of HSMI have been diagnosed on the Atlantic Coast, this suggests that PRV on the East Coast of Canada may not be harmful, or that factors other than the presence of PRV are required to cause the disease.
A survey and characterisation of the local PRV are important in order to know the current distribution of the virus on the East Coast, and the potential for PRV to compromise wild and farmed Atlantic Salmon. This research builds on other studies about PRV.
Date: APR. 2016–MAR. 2020
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Kelly Cove Salmon Ltd.
Project Lead: Nellie Gagné (DFO)
Project Team: Delphine Ditlecadet, Crystal Collette-Belliveau, Jean-René Arseneau, Francis Leblanc, Steven Leadbeater, Philip Byrne (DFO)
Collaborators: Keng Pee Ang (KCS)
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/index-eng.html
Salmon Gill Poxvirus-Like (SGPV-Like): Characterisation, Atlantic Salmon Susceptibility, and Initial Survey in Farmed and Wild Salmon
Poxviruses are large DNA viruses of vertebrates and insects causing disease in many animal species. In the spring of 2006, a new poxvirus, the salmon gill poxvirus (SGPV), was discovered on the gills of salmon suffering from proliferative gill disease (PGD) in freshwater in Northern Norway. Later the same year, this virus was also found on salmon gills at two marine sites in Western Norway where all farms suffered high losses associated with the presence of this virus. Clinical disease symptoms are lethargy, respiratory distress, and mortality.
In 2015, an unknown virus was isolated from tissues sampled from a suspected escaped farmed Atlantic Salmon caught in a river in New Brunswick. Using Next Generation Sequencing, researchers found that there was an 80-90% similarity to Norwegian SGPV, hence “SGPV-like”. The significance of SGPV-like detection is unclear, although there are anecdotal reports that salmon gill diseases of unknown etiology were previously detected on the East Coast of Canada. A survey and characterisation of the local SGPV are needed to determine the extent of the virus distribution on the East Coast, and its potential to compromise the health of salmon.
Date: APR. 2016–MAR. 2020
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Kelly Cove Salmon Ltd. (KCS)
Project Lead: Nellie Gagné (DFO)
Project Team: Delphine Ditlecadet, Valérie Godbout, Jean-René Arseneau, Francis Leblanc, Steven Leadbeater, Philip Byrne (DFO)
Collaborators: Keng Pee Ang (KCS)
Contact: Nellie.Gagne@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/16-1-G-02-eng.html
Description of Oceanographic Conditions within Hermitage Bay, Newfoundland, at Sites with and without the Occurrence of an ISA Outbreak
Infectious Salmon Anemia (ISA) is an important viral disease in the salmon farming industry. The ISA virus (ISAV) has also been found on wild fish and can be transmitted to farmed fish through a number of vectors. ISA outbreaks have been reported in Newfoundland salmon farms between December 2012 and November 2013.
This study aimed to understand how environmental conditions (e.g., temperature, salinity, and dissolved oxygen) at farm sites might favour ISA outbreaks. The link between conditions and ISA outbreaks would help identify other areas at risk of potential outbreaks, assist the aquaculture industry in mitigating the risk, and improve the sustainability of salmon farming.
This project found that ISA outbreaks in Bay d’Espoir occurred in spring/early summer and late autumn and that the general conditions at sites with ISA outbreaks were similar to those without.
At the sites with ISA occurrences, the environmental conditions prior to outbreaks were different between the spring and autumn cases. However, a common range of temperature, salinity, and dissolved oxygen concentration has been observed two weeks prior to the ISA events. A clear link could not be established between the environmental conditions, their variability, and the occurrence of ISA in the outer Bay d’Espoir area.
The occurrence of ISA events in the region could be the result of a combination of various factors including the environmental conditions reported in the present study as well as other processes (e.g., water and particle, ISA virus, movement between sites). Due to the limitation of the data and cases (only four ISA occurrences), further research is required should more cases of ISA events occur in the area.
This project supports the DFO priority of optimal fish health.
Start Date: JUN. 2014–MAR. 2015
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO– ACRDP)
Co-funded by: Cold Ocean Salmon Inc.; Newfoundland Aquaculture Industry Association
Project Lead: Andry Ratsimandresy (DFO)
Project Team: Sebastien Donnet, Steve Snook, Kevin Le Morzadec (DFO)
Collaborators: Julia Bungay (Cold Ocean Salmon Inc.); Miranda Pryor (Newfoundland Aquaculture Industry Association)
Contact: Andry.Ratsimandresy@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/N-14-01-003-eng.html
Investigations into Ulcerative Skin Disease Agents, Moritella viscosa and Tenacibaculum spp. in Atlantic Salmon: Interactions and in Vivo Challenge Development
This project examines two pathogens of concern to the salmon farming industry, Moritella viscosa and Tenacibaculum spp., and their roles in skin ulcerative diseases. The primary focus is to study bacterial isolates of the genus Tenacibaculum which causes a condition called tenacibaculosis. Recent experience in developing immersion challenge models with M. viscosa will be applied to produce a disease challenge model for tenacibaculosis using a Canadian Tenacibaculum isolate. The secondary focus is to study in vitro interactions among M. viscosa and Tenacibaculum isolates and other bacteria associated with skin ulcers. The project focuses on isolates found on the East Coast of Canada in the context of the range of environmental temperatures at which pathogenicity is observed in clinical cases.
The project develops a basis for reproducing tenacibaculosis in Atlantic Salmon within a lab environment, and will provide a challenge model for testing diagnostic techniques for recovery of Tenacibaculum spp. during disease progression. This information could provide the aquaculture industry with improved techniques for early identification of potential causative agents of skin diseases. In addition, a reproducible live challenge model could be used for testing efficacy of treatments and vaccines.
Results of the study can be used by the aquaculture industry for research on genetic selection for disease resistance, help the fish health community apply better diagnostic methodology for confirming and treating ulcerative skin diseases, and improve understanding of naturally occurring diseases that impact wild and farmed salmon.
Date: APR. 2016–JUN. 2018
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Cooke Aquaculture Inc.
Project Lead: Steven Leadbeater (DFO)
Project Team: Allison MacKinnon (Elanco Canada Ltd.); Anthony Manning (RPC)
Collaborators: Leighanne Hawkins (Cooke Aquaculture Inc.)
Contact: Steven.Leadbeater@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/16-1-M-02-eng.html
Cyprinid Herpes Virus 3 (CyHV-3) in Wild Common Carp (Cyprinus carpio L.) in Manitoba, Canada
Lake Manitoba was the site of a 2008 Koi herpes virus disease (KHVD) outbreak due to the presence of the aetiological agent CyHV-3 in resident Common Carp populations. This study was designed to test whether CyHV-3 is enzootic in Common Carp in Lake Winnipeg and Lake Manitoba. Test methods included quantitative PCR (qPCR), enzyme-linked immunosorbent assay (ELISA), duplex conventional PCR, and DNA sequence and phylogenetic analysis. Over the course of four years, carp from Netley Marsh (23.5%; n=17) and Delta Marsh (27.2%; n=235) tested positive for CyHV-3 by qPCR. Virus loads ranged from <5 to 2.9x104 equivalent plasmid copies per µg of DNA (average 7.2x102 copies/µg of DNA ± 3.6x103). Sequence analysis of two variable regions of CyHV-3 DNA located between ORF29 and ORF31 (marker I) and near the 5’ end of ORF133 (marker II) identified a single virus genotype in Manitoba. The marker II allele contained the same novel 13 base pair deletion associated with isolates from East Asia and Southeast Asia. Phylogenetic analysis using the thymidine kinase DNA sequence showed that the Manitoba CyHV-3 isolate shared a closer relationship to isolates from the Asian lineage than with those from the European lineage. Analysis of the ELISA test results suggested that 66.9% (n=239) of the carp sampled were CyHV-3 seropositive. Together, these results suggest that CyHV-3 is now enzootic in Common Carp populations residing in the two largest lakes in Manitoba.
This study examines subsequent changes in carp population dynamics, reports on the distribution, transmission and genetics of CyHV-3 and provides evidence of a carrier state in convalescent carp. The results provide insight into CyHV-3 ecology and are useful to regulators considering the use of CyHV-3 as a biocontrol mechanism for feral Common Carp populations causing widespread water habitat destruction.
Date: JUL. 2009–FEB. 2017
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Sharon Clouthier (DFO)
Project Team: Tamara Schroeder, Melissa Lindsay (DFO): Darius Khambatta (U Manitoba)
Collaborators: Dale Wrubleski, Bob Emery (Ducks Unlimited Canada); Eric Anderson (Ste. Anne, MB)
Contact: Sharon.Clouthier@dfo-mpo.gc.ca

Aerial view of Clair Lake of Delta Marsh with the southern tip of Lake Manitoba evident in the foreground. Photo: Dale Wrubleski (Ducks Unlimited Canada)
Marine Reservoirs of Infectious Agents Associated with Proliferative Gill Disorders in Farmed Salmon
Gill diseases and disorders among Atlantic Salmon raised in seawater net pens are an emergent and important cause of losses. There is a need to better describe the causes, distribution, and possible control of gill diseases, which have been attributed to infections with parasites, bacteria, viruses, as well as exposure to algal blooms, jellyfish, and other non-infectious agents.
This project focuses on associations of viruses and parasites such as Paramoeba perurans and Desmozoon lepeophtherii with proliferative gill disorders (PGD) in order to improve understanding of reservoirs of infections. Infectious causal factors will be studied in populations of wild and farmed salmon sharing common bodies of water to better understand the epidemiology of proliferative gill disorders.
This project will develop knowledge related to the distribution and causes of PGD in British Columbia, which will inform further development of farmed fish health management strategies and improve the understanding of the role of gill disorders in the conservation of wild salmon.
Date: APR. 2016–JUN. 2018
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: BC Salmon Farmers Association (BCSFA)
Project Lead: Simon Jones (DFO)
Project Team: Gary Marty (BC Ministry of Agriculture); Sonja Saksida, Marc Trudel (DFO); Diane Morrison, Sharon DeDominicis (Marine Harvest Canada Limited)
Collaborators: Jeremy Dunn, Joanne Liutkus, Rachel Saraga (BCSFA)
Contact: Simon.Jones@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/16-1-P-02-eng.html
Pathogen Susceptibility of Sockeye Salmon–Phase 1: Infectious Salmon Anemia Virus (ISAV) and Salmon Alphavirus (SPDV or SAV)
In recent years, Sockeye Salmon populations in Canada have been experiencing declines in productivity; of particular note is the Fraser River Sockeye Salmon stock. The reasons for this decline remain speculative but it is thought the susceptibility of Sockeye to pathogens may be a key contributing factor. However, the susceptibility of Sockeye Salmon to Infectious Salmon Anemia Virus (ISAV) or Salmon Alphavirus (SPDV or SAV) has never been tested. These pathogens are known to affect cultured Atlantic Salmon in various salmon producing regions and countries (e.g., ISAV on the east coast of Canada, ISAV and SPDV in Norway).
This project examines the disease resistance or susceptibility of Sockeye Salmon to pathogens known to affect Atlantic Salmon and the potential transmission of disease for a priority list of pathogens (ISAV, SPDV). Additionally, this project compares the viral response to ISAV at a cellular level (e.g., using gill tissue) between Sockeye and Atlantic Salmon, and their long-term immunity in response to exposure. This research is necessary in order to predict the consequences of an introduction or outbreak of these pathogens at an aquaculture site to the health and sustainability of Sockeye Salmon. The outcome of this work will allow Fisheries and Oceans Canada to improve disease surveillance, detection and management strategies, and help to minimize the impacts of pathogens on cultured and wild fish.
Date: JUL. 2014–JUN. 2018
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: Marine Harvest Canada Limited
Project Lead: Nellie Gagné (DFO)
Project Team: Francis Leblanc, Philip Byrne, Steven Leadbeater (DFO)
Collaborators: Diane Morrison (Marine Harvest Canada Limited)
Contact: Nellie.Gagne@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/projects-projets/G-14-01-003-eng.html

Immunostained ISAV infected cells. Photo: Mélanie Roy (DFO)

DFO’s Gulf Biocontainment Unit – Aquatic Animal Health Laboratory in Charlottetown, PEI. Photo: Nellie Gagné (DFO)
The Transfer Potential of Fish Pest and Pathogen from Farmed to Wild Salmon: Stocking Density Effect
Salmon farms can become infected with the Infectious Salmon Anemia Virus (ISAV) from wild fish stocks. Should this occur, farms could potentially become an amplified source of ISAV that could, in turn, impact wild fish. ISAV is transported and dispersed by water currents, and the resulting plumes or ISAV zones may contribute to the transfer of ISAV between farms and to migrating wild salmon that intersect the plumes.
Fisheries and Oceans Canada (DFO) regulators in the Maritimes Region are particularly interested in the role of stocking density in pest and pathogen transfer from existing farms to endangered wild Atlantic Salmon populations. This project aims to produce new experimental information: 1) quantifying the transfer rates of ISAV from farmed Atlantic Salmon; 2) new information on the minimum exposure dose needed to infect naïve salmon; 3) virus shedding rate of infected fish; 4) ultraviolet (UV) and survival of virus in water; as well as 5) models for predicting the potential for waterborne transfer of ISAV from commercial salmon farms under a range of stocking and farm density scenarios. The information will help inform regulators, as well as the aquaculture and recreational salmon industries who are working toward developing and implementing strategies and actions aimed at mitigating the potential for transfer of ISAV from farmed to wild salmon.
Date: APR. 2014–MAR. 2017
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Leads: Fred Page, Nellie Gagné (DFO)
Project Team: Blythe Chang, Francis Leblanc, Steve Leadbeater, Kyle Garver (DFO)
Collaborators: Michael Beattie (NBDAAF); Ian Gardner, Larry Hammel, Crawford Revie, Sophie St. Hilaire, Raphael Vanderstichel (UPEI-AVC)
Contact: Fred.Page@dfo-mpo.gc.ca, Nellie.Gagne@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/parr-prra/projects-projets/2014-m-01-eng.html
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) Assays For Detection of Spring Viremia of Carp Virus–Phases IV and V Diagnostic Validation
Spring viremia of carp virus (SVCV) is the aetiological agent of an aquatic animal disease that is listed as notifiable to the World Organization for Animal Health (OIE) and reportable to the Canadian Food Inspection Agency (CFIA). In this multi-year study, two RT-qPCR tests targeting either the glycoprotein or nucleoprotein gene were designed and optimized for their analytical performance. They were then evaluated along with the virus isolation by cell culture test in a diagnostic validation study to assess their fitness as diagnostic tools for detection of SVCV. Test performance metrics of diagnostic precision were repeatability and reproducibility. Sensitivity and specificity were measured to assess diagnostic accuracy. Estimates of test accuracy, in the absence of a gold standard reference test, were generated using latent class models. Test samples originated from domesticated koi that were either virus free or experimentally infected with the virus. Two tissues, kidney and brain, were evaluated for their relative suitability as target tissues for the RT-qPCR assays. Four laboratories in Canada participated in the precision study. Estimates of test precision and accuracy are being evaluated in the final two phases of the project. The results will determine if any of the three tests are suitable diagnostic tools for detection of SVCV.
Canada is a member of the OIE and the World Trade Organization and as such is obligated, for trade purposes, to implement OIE standards for disease diagnosis. The OIE has SVC on its list of notifiable diseases and in Canada it is currently designated as a CFIA reportable disease. The diagnostic tools developed in this project will be used by member laboratories of the National Aquatic Animal Health Laboratory System to help Canada meet its obligations to protect the health of wild and cultured aquatic animals within the country and globally.
Date: APR. 2013–MAR. 2018
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Sharon Clouthier (DFO)
Project Team: Tamara Schroeder, Shaorong Li, Crystal Collette-Belliveau, Jason Allen, Melissa Lindsay, Sandra Aldous, Philip Byrne (DFO)
Collaborators: Carol McClure (AquaEpi Research); Eric Anderson (Ste. Anne, MB)
Contact: Sharon.Clouthier@dfo-mpo.gc.ca
Marine Reservoirs of Infectious Agents Associated with Proliferative Gill Disorders in Farmed Salmon
Gill diseases contribute to economically important production losses in Atlantic Salmon aquaculture. This project will improve our understanding of reservoirs of infections with infectious agents associated with these disorders. One of these, amoebic gill disease (AGD), was diagnosed for the first time in BC in 2014 and the causative agent, Paramoeba perurans, has been detected in BC. Another agent that is associated with proliferative gill inflammation (PGI) in Europe, the microsporidian, Desmozoon lepeophtherii, has been detected in BC and Washington State. Several cases of a PGI-like condition were reported in farmed salmon in BC in 2015. This project capitalizes on an opportunity to benefit from a unique set of samples; juvenile salmon collected by industry as part of their sea lice certification program. Specifically, these samples will be examined for evidence of their involvement as reservoirs of infection with agents associated with proliferative gill disorders in farmed salmon.
The objectives of this project are to:
- determine distribution of P. perurans and D. lepeophtherii in wild Pacific Salmon and salmon lice collected in proximity to marine netpens;
- describe the occurrence of proliferative gill lesions in wild fish;
- characterize the genomic sequence of BC variants of P. perurans and D. lepeophtherii; and
- conduct laboratory transmission studies to identify and quantify (host and environmental) parameters surrounding transmission of the causative agent between candidate reservoir species and Atlantic Salmon.
These objectives reflect the knowledge gaps concerning PGI and causal agents in farmed salmon in BC. The key anticipated project outcome will be knowledge related to the distribution and causes of PGI in BC. This knowledge will inform further development of farmed fish health management strategies.
Date: APR. 2016–MAR. 2018
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Lead: Simon Jones (DFO)
Project Team: Anne-Marie Flores, Sonja Saksida (DFO)
Collaborators: Diane Morrison, Sharon DeDominicis (Marine Harvest Canada Limited); Marc Trudel (DFO)
Contact: Simon.Jones@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/parr-prra/index-eng.html
Epidemiological Analysis and Modelling of Aquatic Pathogens
One of the big questions concerning disease processes of aquatic pathogens is what factors influence pathogen prevalence in wild and cultured fish populations. The prevalence and intensity of pathogens can vary annually within and between fish stocks, yet the drivers behind such annual fluctuations remain unresolved. Some fish populations can be free of virus one year, yet be greater than 80% positive in subsequent years. The occurrence of such dramatic “on and off periods” begs the questions “Why now?” and “Why was there an absence of virus in previous years?”
To better understand variations in aquatic pathogen prevalence in wild and farmed populations, this project will use epidemiological analytical approaches to study the patterns of aquatic pathogens in wild and cultured fish populations with the goal of identifying the factors that influence the occurrence of pathogens and disease.
To help understand the main drivers of pathogen epidemiology in aquatic systems, this project will: 1) gather and assemble a comprehensive dataset of fish health analyses and salmon population metrics across multiple river systems; 2) examine and compare annual pathogen prevalence among wild sockeye populations from multiple river systems; and 3) explore host, pathogen and environmental factors for potential correlates with pathogen variability in wild sockeye populations.
Date: APR. 2016–MAR. 2018
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Lead: Kyle Garver (DFO)
Project Team: Christine MacWilliams (DFO)
Collaborators: Carl Ribble (Centre for Coastal Health Society)
Contact: Kyle.Garver@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/parr-prra/projects-projets/2016-P-02-eng.html
Diagnostic Validation of Three Test Methods for Detection of Cyprinid Herpesvirus 3
Cyprinid herpesvirus 3 (CyHV-3) is the aetiological agent of koi herpes virus disease (KHVD) in Common Carp (Cyprinus carpio L.) and Koi. The disease is notifiable to the World Organization for Animal Health (OIE). Three tests [quantitative polymerase chain reaction (qPCR), conventional PCR (cPCR), and virus isolation by cell culture (VI)] were validated to assess their fitness as diagnostic tools for detection of CyHV-3. Test performance metrics of diagnostic accuracy were sensitivity (DSe) and specificity (DSp). Repeatability and reproducibility were measured to assess diagnostic precision. Estimates of test accuracy, in the absence of a gold standard reference test, were generated using latent class models. The test samples originated from wild Common Carp naturally exposed to CyHV-3 or domesticated Koi, either virus-free or experimentally infected with the virus. Three laboratories in Canada participated in the precision study. Moderate to high repeatability (81% to 99%) and reproducibility (72% to 97%) were observed for the qPCR and cPCR tests. The lack of agreement observed between some of the PCR test pair results was attributed to cross contamination of samples with CyHV-3 nucleic acid. Accuracy estimates for the PCR tests were 99% for DSe and 93% for DSp. Poor precision was observed for the virus isolation test (4% to 95%). Accuracy estimates for VI/qPCR were 90% for DSe and 88% for DSp. Collectively, the results show that the CyHV-3 qPCR test is a suitable tool for surveillance, presumptive diagnosis, and certification of individuals or populations as CyHV-3 free.
Canada is a member of the OIE and the World Trade Organization and as such is obligated, for trade purposes, to implement OIE standards for disease diagnosis. The OIE has KHVD on its list of notifiable diseases and in Canada it is designated as reportable to the Canadian Food Inspection Agency. The diagnostic tools developed in this project for detection of CyHV-3 will be used by member laboratories of the National Aquatic Animal Health Laboratory System (NAAHLS) to help Canada meet its obligations to protect the health of wild and cultured aquatic animals within the country and globally.
Date: JUL. 2009–NOV. 2016
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Sharon Clouthier (DFO)
Project Team: Tamara Schroeder, Megan Desai, Laura Hawley, Sunita Khatkar, Melissa Lindsay, Geoff Lowe, Jon Richard (DFO)
Collaborators: Carol McClure (AquaEpi Research); Eric Anderson (Ste. Anne, MB)
Contact: Sharon.Clouthier@dfo-mpo.gc.ca
Physiological Consequences of Piscine Orthoreovirus (PRV) Infection of Atlantic and Pacific Salmonids
Piscine Orthoreovirus (PRV) commonly occurs in healthy salmon which are asymptomatic of heart and skeletal muscle inflammation (HSMI) disease. The capacity of PRV to establish long-term infections and the potential association with the HSMI disease in fish has raised concerns towards its ability to impact salmon physiology and overall fish health. As such, the role of PRV in HSMI continues to be studied.
This project aims to determine if PRV infections have physiological consequences and whether the virus can directly impact salmon health. This laboratory study addresses these questions by evaluating the cardiorespiratory performance (swimming ability) of PRV-infected Atlantic and Sockeye Salmon in comparison to uninfected controls. Oxygen binding potential and maximum oxygen consumption will be measured and compared between infected and non-infected individuals.
This research builds on other studies about PRV. The outcome of this study will provide a better understanding of whether PRV infections are associated with physiological consequences. Results of the study will inform Fisheries and Oceans Canada management strategies for PRV.
Date: AUG. 2016–JUN. 2018
Funded by: DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP)
Co-Funded by: BC Salmon Farmers Association (BCSFA)
Project Lead: Kyle Garver (DFO)
Project Team: Mark Polinski (NSERC); Anthony Farrell, Colin Brauner (UBC)
Collaborators: Jeremy Dunn (BCSFA)
Contact: Kyle.Garver@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/acrdp-pcrda/index-eng.html
Does Infection with Piscine Reovirus (PRV) Affect How Salmon Respond to Challenge with Infectious Hematopoietic Necrosis Virus (IHNV)?
Piscine reovirus (PRV) is a virus common in wild and farmed salmon in BC and likely establishes long term infections in its host. It is inevitable that mixed infections of PRV and known pathogens will occur. One pathogen that is in the same geographical area with PRV is the infectious hematopoietic necrosis virus (IHNV) which occurs naturally in Pacific Northwest waters. With the appropriate virus strain, host condition, and environmental parameters, IHNV can cause an acute disease (IHN) in all five species of Pacific salmon, Atlantic Salmon, and Rainbow Trout.
This project seeks to examine the consequences of viral co-infections of PRV and IHNV in Sockeye Salmon. The study will address how Sockeye Salmon hosts, infected with viruses with no or low pathogenic effect, respond to challenge with other viruses. Challenge trials will be used to examine IHN disease progression in naïve (never before infected) and PRV-infected Sockeye Salmon. This challenge trial will determine if there are differences between groups in morbidity associated with IHNV challenge, and to generate biological samples for gene and microRNA expression studies. This research will help to determine what, if any, additional risk is posed to wild and/or farmed fish due to changes in their ability to respond to exposure to IHNV, thereby contributing to the knowledge base important for the sustainable management of the salmon aquaculture industry.
Date: APR. 2014–MAR. 2017
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Leads: Stewart Johnson, Kyle Garver (DFO)
Project Team: Jon Richards, Julia Bradshaw (DFO)
Collaborators: Matthew Rise (MUN); Rune Adnreassen (Oslo and Akershus University College of Applied Sciences)
Contact: Stewart.Johnson@dfo-mpo.gc.ca, Kyle.Garver@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/parr-prra/projects-projets/2014-P-13-eng.html
Addressing the Question of Sturgeon Nucleo-Cytoplasmic Large DNA Virus (sNCLDV) Systematics
Sturgeon nucleo-cytoplasmic large DNA viruses (NCLDV) are epitheliotropic viruses associated with mortality in nine sturgeon species in North America and Europe. White Sturgeon iridovirus (WSIV), a member of this group, causes a disease that is reportable to the Canadian Food Inspection Agency (CFIA). When the virus was originally described in the 1990’s, it was classified as a member of the Iridoviridae family based on its morphological characteristics. In 2009, Namao virus (NV) was discovered in populations of moribund lake sturgeon in Manitoba, Canada and found to be genetically related to WSIV. This study was undertaken to investigate the evolutionary relationship of NV to other NCLDVs. The goal was to resolve the systematics of NV within the proposed order Megavirales given that our initial phylogenetic analyses with the major capsid protein sequence of five sNCLDVs suggested that the taxa formed a monophyletic group outside of the Iridoviridae family. NV genome sequence has been determined for three non-overlapping contigs with an overall length of 288.4 kb. A set of 10 orthologous genes present in the genomes of all NCLDVs are being used to build a series of phylogenetic trees for each gene and to construct a consensus tree. A total of 40 taxa representing genera from six virus families have been chosen. The sequences for each gene will be aligned and the evolutionary relationship of NV with other NCLDVs will be examined using multiple phylogenetic software packages including coalescent analyses with the BEAST suite of programs and maximum likelihood analyses using MEGA.
The study has been designed so that the phylogeny outcomes would be taxonomically informative to the International Committee on Taxonomy of Viruses. The results of this research may impact how WSIV and, by extension, other sNCLDVs are regulated by the CFIA and by regulators of national aquatic animal health programs in Europe and the United States.
Date: JAN. 2012–JUN. 2017
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Co-Funded by: Manitoba Hydro
Project Lead: Sharon Clouthier (DFO)
Collaborators: Rachel Breyta, Gael Kurath (US Geological Survey–Western Fisheries Research Center); Eric Anderson (Ste. Anne, MB)
Contact: Sharon.Clouthier@dfo-mpo.gc.ca
Stage 1 Validation of Real-Time PCR (qPCR) Assay for the Detection of Bonamia spp.
Assays for this pathogen vary in their ability to detect one or both of pathogens regulated by the Canadian Food Inspection Agency (CFIA); Bonamia ostreae and Bonamia exitiosa. The objectives of this project are: 1) to complete a World Organization for Animal Health (OIE) stage 1 validation (determine the operating range, analytical specificity and sensitivity, repeatability and characterization of inhibition) for a modified version of a previously published real-time PCR (qPCR) assay capable of detecting both of these pathogens; 2) the design and characterization of an artificial internal positive control (G-block) for use with the assay; and 3) the verification of a previously published conventional, PCR based, confirmatory assay which, along with Sanger based DNA sequencing, distinguishes between these species. The results of this validation and control development will be described in a validation dossier prepared for the National Aquatic Animal Health Program (NAAHP).
1) A sybr-green based qPCR (based on the work of Hill et al., 2010 and 2014), capable of distinguishing between Bonamia spp. using melt-curve analysis, has been validated to OIE stage 1 requirements. 2) An artificial internal positive control (IPC) has been created that is distinguishable from both ‘wild-type’ infections and demonstrates the same reaction kinetics as natural infections of both B. ostreae and B. exitiosa. 3) An existing conventional PCR confirmatory assay (based on the work of Cochennec et al., 2000) was optimized and tested on B. ostreae, B. exitiosa and the IPC and was able to distinguish between the two pathogens but did not amplify the IPC (this is by design to prevent false ‘detected’ results). However, it should be noted that the confirmatory assay was found to be less sensitive than the screening assay.
Date: APR. 2016–MAR. 2017
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Leads: Cathryn Abbott, Geoff Lowe (DFO)
Contact: Cathryn.Abbott@dfo-mpo.gc.ca
Development and Diagnostic Validation of a qPCR Assay to Detect Mikrocytos mackini and its Application in Understanding the Transmission Risks and General Biology of the Pathogen
This project will finalize the validation process and produce a manuscript and validation dossier for the recently developed qPCR diagnostic assay intended for routine shellfish screening. The validated qPCR assay provided field data for the project described below and publication of the data and a validation dossier is in progress. Once completed, this will provide the international aquatic animal health community with a fully validated and characterized assay for the detection of this important shellfish pathogen.
The second part of this project will use the qPCR described above to: 1) determine the environmental stability of Mikrocytos mackini; 2) assess the ability and practicality for environmental diagnostic screening; 3) map M. mackini infections within host tissues so as to better understand routes of transmission that can be used to identify/manage disease outbreaks; and 4) lead to a publication and dissemination of these findings that will aid in Canadian and international management of M. mackini (and likely, by extension, other related parasites).
In this project we expanded the utility of this assay to detect M. mackini from environmental samples and demonstrated that M. mackini was shed into culture water from infected oysters following experimental injection. Further investigation revealed limited environmental stability of M. mackini, as most material became non-infectious after a period of 24 h in seawater. Mapping of infections within host tissues revealed that exposure to M. mackini of naïve oysters in an aquatic environment demonstrated gill associated parasites within one day following exposure. These results indicate that in natural environs, M. mackini may be shed into the water by infected oysters but the limited environmental stability of the parasite likely restricts its spread causing focal infection dynamics within a host population.
Date: APR. 2016–MAR. 2017
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Leads: Cathryn Abbott, Mark Polinski (DFO)
Project Team: Gary Meyer, Geoff Lowe, Eliah Kim (DFO)
Contact: Cathryn.Abbott@dfo-mpo.gc.ca
Effects of Pooling Animals on the Probability of Detection: MSX and ISAV
Although pooling of animals is not currently used in the National Aquatic Animal Health Laboratory System (NAAHLS), it is often discussed, as it has the advantage of processing multiple animals at once. This can greatly reduce the processing time and increase the throughput of a laboratory. By pooling two animals, one doubles the throughput, and pooling more than two animals is possible. However, there is a trade-off to pooling; when only one animal is infected, and the animal is infected at a level near the limit of detection, the dilution effect of the pooling process reduces the probability of detection. This reduction in diagnostic sensitivity needs to be carefully evaluated at the bench level.
This project will provide: 1) a set of data (probability of detection) of a single positive animal in a pool of two, three, four, or five negative animals, for DNA extraction methods (with MSX as a pathogen) and RNA extraction methods (ISAV pathogen); and 2) in practice, the data produced in this project should be transferable directly to other assays where pooling is desired.
Knowing the impact of pooling, clients will be able to decide if the decline in sensitivity is worth the trade-off, based on their reason for testing and the current disease prevalence.
Date: APR. 2015–MAR. 2016
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Nellie Gagné (DFO)
Project Team: Francis Leblanc, Crystal Collette-Belliveau, Valérie Godbout (DFO)
Contact: Nellie.Gagne@dfo-mpo.gc.ca
Identification of Vectors of MSX to Support Introductions and Transfers Decisions Related to Inter-Provincial Movements of Mussels: Is Mussel Intra-Valvular Liquid a Vector for MSX Transmission?
Multinucleated sphere “X” (MSX) is an infectious disease that causes heavy mortalities in American Oysters (Crassostrea virginica) although it does not affect human health. The transfer of live shellfish, such as mussels, are regulated under Section 56 of the Fisheries (General) Regulations and are reviewed on a case by case basis by the DFO Introductions and Transfer Committees (ITC) of the receiving Province. Prior DFO analysis of mussels collected from heavily infected MSX positive areas of the Bras d’Or Lakes, and from MSX positive areas outside of the Lakes, had not detected MSX in the soft tissues/intra-valvular liquid. In the absence of information, epifauna on mussel lines was considered potentially positive for MSX and mitigation was recommended by the ITCs. In this project, targeted sampling of wild mussels and epifauna in an area with MSX infected wild oysters resulted in the detection of MSX by Real Time Polymerase Chain Reaction (qPCR) but the disease was not visually detected histologically. These results support industry concerns that although mussels are not believed to be a host of MSX, a life stage of the parasite may be present in the intra-valvular fluid of the mussels or the intermediate host may be found within the associated epifauna. Therefore, based on the results of this project, continued mitigation to reduce the risk of the inadvertent spread of MSX should be considered during risk assessments of industry activities.
Date: APR. 2013–MAR. 2015
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Lead: Mary Stephenson (DFO)
Project Team: Nellie Gagné, Marie-Line Cournoyer (DFO)
Collaborators: Andrew Bagnall (NSDFA)
Contact: Mary.Stephenson@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/parr-prra/index-eng.html
Effect of Water Temperature on the Immune Response of American Lobster (Homarus americanus) Experimentally Infected with White Spot Syndrome Virus
Water temperature influences many basic biological functions of the American Lobster (Homarus americanus); yet, little is known about the effects of temperature on the immune response of H. americanus. White Spot Syndrome Virus (WSSV) is one of the largest impediments to shrimp aquaculture worldwide; there are no reports of naturally occurring WSSV infection in lobster. The World Organization for Animal Health (OIE) lists WSSV as a notifiable disease with the potential to infect all crustacean decapods.
This project utilized a previously established infection challenge model to investigate the constraints imposed by a range of temperatures (10°C, 15°C, 17.5°C, and 20°C) on the clinical, tissue, and molecular immune responses of American Lobster experimentally infected with WSSV. The infection model utilized intramuscular injection inoculation of WSSV. This study found that WSSV amplification was associated with increased water temperatures (17°C and 20°C). Significant differences in total haemocyte concentration between infected and control H. americanus at 17.5°C and 20°C, but not at 10°C and 15°C. This suggests a decrease in clinical condition of WSSV infected H. americanus at warmer temperatures. Light and transmission electron microscopy were used to elucidate tissue changes in the host and confirmed the antennal gland as a key target tissue for WSSV. A lobster specific microarray identified 717 significantly differentially expressed genes between infected and control animals at the various temperatures. Microarray results highlighted differential expression of various temperature and immune-related genes. At 20°C, H. americanus appears to produce a targeted immune response to injected WSSV.
This project used a range of diagnostic tools and experimental temperatures in order to broaden our understanding of how temperature influences host pathogen interactions in the economically important crustacean species, the American Lobster (H. americanus). Although this project and other work done involving DFO and the Lobster Science Centre collaboration suggests that WSSV is not a threat to lobster, the use of the WSSV experimental infection model has been useful for enabling a comparative examination of the lobster immune response to different kinds of pathogens (bacterial, protozoal, and viral).
Date: SEP. 2014–SEP. 2016
Funded by: University of Prince Edward Island–Atlantic Veterinary College (UPEI–AVC)
Co-Funded by: Federal Student Work Experience Program (FSWEP); DFO–Centre for Aquatic Animal Health Research and Diagnostics DFO–CAAHRD)
Project Leads: Philip Byrne (DFO); Spencer Greenwood (UPEI-AVC)
Project Team: Louise-Marie Roux (UPEI–AVC; DFO)
Collaborators: Fraser Clark, Mark Fast, Glenda Wright, Dorota Wadowsk (UPEI-AVC)
Contact: LRoux@upei.ca

Heat map with hierarchical clustering of significantly differentially expressed genes identified between infected Homarus americanus, two-week post inoculation at 10°C, 15°C, 17.5°C, and 20°C.
Red represents a threefold or greater decrease in gene expression whereas green represents a threefold or greater increase in gene expression.

Louise-Marie Roux in the Gulf Biocontainment Unit (DFO) facility collecting a haemolymph sample from American Lobster (Homarus americanus). Photo: Phil Byrne (DFO)

Transmission Electron Micrograph of WSSV infected Homarus americanus antennal gland, two-weeks post inoculation at 17.5°C.
Hypertrophic nuclei showing marginated host chromatin and containing WSSV viral particles within the nucleus identified by an electron dense nucleocapsid center and a trilaminar envelope. Virions ranged from 200-350 nm in length. Nucleus, apical microvilli, mitochondria (mt), and vacuole (va) labelled.
Developing a Genomics Tool (FIT-Chip) for In-Season Information on Salmon Health
Climate change, genetic challenges, ecological inconsistency, and disease can all be listed as potential stressors to wild populations of salmon on Canada’s West Coast. This project plans to address the physical states of wild populations through the development of a series of “salmon FIT-CHIPs” which will be used as a predictor of salmon condition. The system offers extreme flexibility, allowing biological markers to be easily customized for stress or other conditional states to different life-history stages, environments, and in different tissues. This tool will provide the first in-season method to incorporate information on salmon condition into fisheries management decision making.
A text search will be conducted on abstracts or full papers for key protein (biomarkers) word associations that have been linked to a given stressor. The project will then continue by data-mining the huge in-house microarray databases produced from dozens of studies that encompass more than 4,900 microarrays and three salmon species (Chinook, Sockeye, and Coho) as well as over 60 stress-challenge microarray studies conducted across a broad range of salmon species available in public databases. Tests will be developed that should work across all three species, and preferentially across Atlantic Salmon and Rainbow Trout as well. In all, it is anticipated that five FIT-CHIPS will be developed through this project.
The ultimate aim is to discover biomarkers predictive of the presence of specific stressors or conditions and to translate these into a tool (FIT-CHIP) that can inexpensively and rapidly identify the presence of multiple stressors or conditions in a group of fish at once.
Date: APR. 2014–MAR. 2017
Funded by: DFO–Genomics Research and Development Initiative (DFO–GRDI)
Co-Funded by: Genome BC
Project Lead: Kristi Miller-Saunders (DFO)
Project Team: Norma Ginther, Tobi Ming, Colin Wallace (DFO)
Collaborators: Paul Pavlidis (UBC)
Contact: Kristi.Saunders@dfo-mpo.gc.ca
Website: Developing a genomics tool (FIT-CHIP) for in-season information on Salmon health
Susceptibility of Sockeye Salmon to Viral Hemorrhagic Septicemia Virus
As part of the sustainable management of finfish aquaculture, there are regulatory requirements that are designed to minimize the transfer of pathogens from farmed finfish to wild fish. However, finfish cultured in ocean net pens have the potential to be exposed to naturally occurring pathogens and to transfer pathogens to wild fish. Viral Hemorrhagic Septicemia Virus (VHSV) has been identified in farmed Atlantic Salmon, Salmo salar, in British Columbia. VHSV is a naturally occurring pathogen in British Columbia and is a cause of serious disease in wild Pacific Herring, Clupea pallasii. Pacific Herring are known to enter and remain in Atlantic Salmon net pens, thereby increasing the potential for the virus to be transferred to farmed Atlantic Salmon. Although the occurrence of VHSV in farmed salmon is rarely accompanied with significant disease and mortality, experimental studies have shown that the VHS virus can persist in the tissue of farmed Atlantic Salmon. This causes concern for potential host adaptation and viral spillback to fish sharing the marine environment.
This study is investigating what effect, if any, the occurrence of VHSV in farmed Atlantic Salmon may have on wild Sockeye Salmon. Previous studies have shown that Coho, Chinook, and Chum Salmon are natural hosts of VHSV, but less is known about the susceptibility of Sockeye Salmon to VHSV. To address this knowledge gap, this project will determine the susceptibility of Sockeye Salmon smolts to VHSV using laboratory exposure studies. The results of this study will contribute to the development of adaptive management strategies in British Columbia aimed at minimizing the transfer of pathogens between farmed Atlantic Salmon and wild Pacific salmon.
Date: APR. 2015–MAR. 2017
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Lead: Kyle Garver (DFO)
Project Team: Paul Hershberger (United States Geological Survey); Jon Richard (DFO)
Contact: Kyle.Garver@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/parr-prra/projects-projets/2015-P-01-eng.html
Epidemiology of Ulcer Disease in Salt Water Atlantic Salmon
Working with the aquaculture industry, we investigated the pattern of ulcer disease occurrence in salt water Atlantic Salmon. We also conducted a laboratory trial to: 1) evaluate waterborne transmission of Moritella viscosa; 2) describe the progression of the disease within a population; and 3) determine if skin ulcers could be induced from exposure to the extracellular toxins produced by M. viscosa.
To effectively treat ulcer disease, our research suggests that treatments would likely have to be applied very early in the disease process because fish stop eating once they develop the ulcer. Further, it is relatively difficult to transmit M. viscosa via water, and the lesion associated with M. viscosa can be induced by exposure to the extracellular toxins of M. viscosa. The latter may explain why fish have a strong tissue response when exposed to M. viscosa, but bacteria are difficult to identify on histological evaluation.
Date: MAR. 2015–OCT. 2016
Funded by: Natural Sciences and Engineering Research Council (NSERC)
Project Lead: Sophie St-Hilaire (UPEI)
Project Team: Brett MacKinnon (UPEI)
Contact: ssthilaire@upei.ca
Infectious Salmon Anemia Virus Susceptibility and Health Status of Wild Versus Farmed Atlantic Salmon: A Comparative Study
There are concerns about the potential for interaction between farmed and wild Atlantic Salmon in areas where they coexist. The Committee on the Status of Endangered Wildlife in Canada (COSEWIC) has designated wild Atlantic Salmon populations in Atlantic Canada as threatened or endangered. While the health status and disease resistance of farmed Atlantic Salmon is well documented, the information for wild Atlantic Salmon is less abundant. For example, Infectious Salmon Anemia Virus (ISAV) remains a recurrent problem for cultured salmon in Atlantic Canada, with outbreaks detected in Nova Scotia and Newfoundland since 2012. There are knowledge gaps regarding the prevalence of this virus in wild populations as well as the potential transmission between wild and cultured stocks. As such, the aim of this project is to use in vivo disease challenges and next generation sequencing technologies to compare the susceptibility of wild Atlantic Salmon stocks (Saint John River, Inner Bay of Fundy, Miramichi, Margaree River stocks) and cultured stocks (Saint John River origin) to diseases such as ISAV and others. This study will examine genetic differences and measure immune responses, which could potentially explain any observed differences in susceptibility of wild and cultured salmon to ISAV. It will also provide a general assessment of the health status of wild stocks. The evolution rate of ISAV will be examined by looking at full ISAV sequences in tissue of farmed and wild Atlantic Salmon sampled throughout the course of the in vivo challenges. This project will address the knowledge gap regarding the susceptibility of wild Atlantic Salmon compared to farmed Atlantic Salmon to ISAV.
Date: APR. 2014–MAR. 2017
Funded by: DFO–Program for Aquaculture Regulatory Research (DFO–PARR)
Project Lead: Nellie Gagné (DFO)
Project Team: Francis LeBlanc, Mark Laflamme, Gérald Chaput, Steven Leadbeater, John Whitelaw (DFO)
Collaborators: Mark Hambrook (Miramichi Salmon Association)
Contact: Nellie.Gagne@dfo-mpo.gc.ca
Website: www.dfo-mpo.gc.ca/aquaculture/rp-pr/parr-prra/projects-projets/2014-G-11-eng.html

Atlantic Salmon from the Miramichi River. Photo: Steven Leadbeater (DFO)
Rapid Detection of Replicating Infectious Salmon Anemia Virus (ISAV)
In the diagnostic lab, the traditional methods of virus detection are based on the culture or isolation of the virus. These methods are quite sensitive, and very reliable. They are based on the observation that many viruses will replicate in cells, and eventually rupture the cells. These burst cells can be seen under a microscope, and thus burst cells equal the presence of a replicating (live) virus. However, this observation can take many days. Emerging, modern methods are based on the detection of virus nucleic acids through amplification by polymerase chain reaction (PCR). These methods are ultra-sensitive, and very fast, but they are only indirectly detecting virus. In other words, it is debatable whether what is being detected can be associated to a replicating virus or just “dead” viruses.
Our rapid detection protocol uses both traditional and modern techniques in a “best of both worlds” approach. Test material is put in culture with cells, and a molecular PCR assay is done at that time as well. The PCR assays determines the presence of all viral particles, whether they are “alive” or “dead”. A few days later, a second PCR is done on the cell culture. If the number of viral particles has increased, this can only be explained by the fact that viruses were replicating.
The combined use of two PCR reactions along with the infection of cell lines can identify replicating virus an average of 25 days faster than using the current virus isolation protocol alone. Currently, the detection of the ISA virus can take over 40 days. Given the immense impact this virus can have on cultured fish, this is simply too long. Our protocol can provide decision makers with a much earlier warning.
Date: MAR. 2014–MAR. 2016
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Mark LaFlamme (DFO)
Project Team: Jean-René Arseneau (DFO)
Contact: Mark.LaFlamme@dfo-mpo.gc.ca
Disease and Parasite Resistance Genomics in a Commercial Strain of North American Atlantic Salmon
Bacterial kidney disease (BKD), infectious salmon anemia (ISA), and salmon lice (Lepeophtheirus salmonis) are economically important pathogens to Atlantic Salmon aquaculture on the east coast of Canada. This research project aims to detect molecular markers (SNPs) that are associated with pre-existing genetic resistance in the Saint John River strain of Atlantic Salmon for BKD, ISA, and L. salmonis.
Smolts from the breeding nucleus are exposed to each pathogen in separate challenges to quantify the level of resistance of siblings of potential broodstock. Challenged fish were genotyped using a custom 50,000 (50K) SNP chip designed specifically for North American Atlantic Salmon. Analysis of the SNP genotypes, resistance phenotypes, and the pedigree information can identify SNP markers associated with resistance to these pathogens. These markers can then be incorporated into breeding programs to develop Atlantic Salmon lines that have an increased resistance to BKD, ISA, and L. salmonis.
Date: APR. 2014–MAR. 2018
Funded by: Genome Canada
Co-Funded by: Cooke Aquaculture Inc.; Genome Atlantic; Ontario Genomics; DFO–Aquaculture Collaborative Research and Development Program (DFO–ACRDP); Industrial Research Assistance Program (NRC); Atlantic Canada Opportunities Agency (ACOA)
Project Lead: Elizabeth Boulding (U Guelph)
Project Team: Melissa Holborn, Larry Schaeffer, Jane Tosh (U Guelph); Keng Pee Ang, Jake Elliott, Frank Powell (Cooke Aquaculture Inc.); Steven Leadbeater (DFO); Brian Glebe (Genome Atlantic)
Collaborators: Norwegian University of Life Sciences
Contact: boulding@uoguelph.ca
Website: https://www.uoguelph.ca/ib/boulding

Melissa Holborn (U Guelph) counting salmon lice on Atlantic Salmon smolts. Photo: Cindy Hawthorne (DFO)
Development of Artificial Reference Material for Assessing IHNV and VHSV RT-qPCR Assays
Reference materials, commonly referred to as controls or standards, are substances with extremely well defined properties used in the assessment of diagnostic methods. The Virology laboratory at DFO’s Pacific Biological Station (PBS) has been experimenting with various control materials that can be used to accurately assess the validity of IHNV and VHSV molecular diagnostic tests. The Virology laboratory has initiated the use of an artificial positive control (APC) for monitoring test performance. APCs have an advantage over traditional controls due to the fact that they are unrelated to the target pathogen and can be easily differentiated from each other, facilitating the identification of false positive results.
Results from small scale pilot studies proved promising for proficiency testing of IHNV and VHSV RT-qPCR in that the artificial transcripts mimicked the biological target, could be accurately quantified, and present extremely low false positive risk due to its unique identity. However, uncertainties remain concerning the large scale production of such panels. In particular, what are the best manufacturing methods suitable for generating large batches of homogenous aliquots? What preservation methods are best for large batches of proficiency panels and what is the expected stability? The aims of this study was threefold: 1) establish procedures for generating large batches of IHNV and VHSV artificial reference material for use as standards, controls, and proficiency testing; 2) identify optimal storage practices and best before dates of bulk reference materials; and 3) develop characterization procedures for IHNV and VHSV bulk reference materials.
Artificial positive controls are now an integral component of laboratory quality management systems and a requirement of ISO 17025. By using APCs, the NAAHLS eliminates the risk of false positives. Reference materials are distributed among the NAAHLS as needed to ensure compliance with national protocols and certify testing competency.
Date: SEP. 2014–MAR. 2015
Funded by: DFO–Centre for Aquatic Animal Health Research and Diagnostics (DFO–CAAHRD)
Project Lead: Kyle Garver (DFO)
Project Team: Laura Hawley, Jon Richard (DFO)
Contact: Kyle.Garver@dfo-mpo.gc.ca
- Date modified: